The biochemical analysis of Hibiscus sabdariffa extracts’ antioxidant properties and their function as a natural source of phenolics and anthocyanins
التحليل الكيميائي الحيوي لمستخلصات الكركديه ودورها كمصدر طبيعي للفينولات والأنثوسيانين وخصائصها المضادة للأكسدة
Ahmed A. Zuhaira*1, Saba Abd Al-Mutleb Hamood**1, Muthik Abd Muslim Guda***2
1 Department of Medical physics, Faculty of Medical Sciences, Jabir Ibn Hayyan University for Medical and Pharmaceutical Sciences, Najaf, Iraq.
2 Department of Ecology, Faculty of Sciences, Kufa University, Najaf, Iraq
*ahmed.a.abdulwahid@jmu.edu.iq ,
**saba.a.hamood@jmu.edu.iq
***meethakha.almithhachi@uokufa.edu.iq
DOI: https://doi.org/10.53796/hnsj610/45
Arabic Scientific Research Identifier: https://arsri.org/10000/610/45
Volume (6) Issue (10). Pages: 747 - 758
Received at: 2025-09-07 | Accepted at: 2025-09-15 | Published at: 2025-10-01
Abstract: This study aimed toward assessing the antioxidant properties of Hibiscus sabdariffa L. extracts by assessing the concentrations of phenolic compounds and anthocyanins, which are natural bioactive chemicals with potential health benefits. Dried flower specimens were procured from local market places in Najaf, Iraq, and extracted via an eco-friendly ultrasonic-assisted extraction technique. Qualitative and quantitative studies of phenolic and anthocyanin ingredients have been done employing. The use of HPLC for high-performance liquid chromatography. According to the results, there was proof of seven main phenolic compounds, with rutin reaching 145.6 ppm and quercetin reaching 123.5 ppm. and rosmarinic acid (98.7 ppm) being the most prominent. Additionally, three principal anthocyanins were identified: malvidin (91.4 ppm), cyanidin (78.7 ppm), and delphinidin (71.9 ppm). The bioassays demonstrated that the extract revealed potent antioxidant activity surpassing that of vitamin C, as evidenced by the DPPH assay with a 96% inhibition rate. The extract demonstrated significant hydrogen peroxide scavenging capacity and ferric ion reduction power, revealing its significant abilities in alleviating. The process of reducing oxidative stress and neutralizing free radicals. The extract's remarkable biological activity is enhanced by the presence of various bioactive compounds, including flavonoids, anthocyanins, phenols, as confirmed by phytochemical screening. According to the research, Hibiscus sabdariffa is a significant natural source of phenolic and anthocyanin derivatives with potent antioxidant properties, making it a safe and useful natural antioxidant for use in the food and pharmaceutical industries.
Keywords: Hibiscus sabdariffa L., roselle, phenolic compounds, anthocyanins, antioxidant activity, HPLC analysis, phytochemical screening, DPPH assay, natural antioxidants.
المستخلص: هدفت هذه الدراسة إلى تقييم الخصائص المضادة للأكسدة لمستخلصات نبات الكركديه (Hibiscus sabdariffa L.) من خلال قياس تراكيز المركبات الفينولية والأنثوسيانينات، وهي مركبات حيوية طبيعية فعالة تمتلك فوائد صحية محتملة. تم الحصول على عينات من الأزهار المجففة من الأسواق المحلية في النجف، العراق، وتم استخلاصها باستخدام تقنية صديقة للبيئة تعتمد على الاستخلاص بالموجات فوق الصوتية. أُجريت الدراسات النوعية والكمية للمكونات الفينولية والأنثوسيانينية باستخدام الكروماتوغرافيا السائلة عالية الأداء (HPLC). أظهرت النتائج وجود سبعة مركبات فينولية رئيسية، كان من أبرزها الروتين (145.6 جزء في المليون) والكويرسيتين (123.5 جزء في المليون) وحمض الروزمارينيك (98.7 جزء في المليون). كما تم تحديد ثلاثة أنواع رئيسية من الأنثوسيانينات، وهي: المالفيدين (91.4 جزء في المليون)، السيانيدين (78.7 جزء في المليون)، والدلفينيدين (71.9 جزء في المليون). أظهرت الاختبارات البيولوجية أن المستخلص يتمتع بنشاط مضاد قوي للأكسدة يفوق نشاط فيتامين C، وذلك وفقًا لاختبار DPPH الذي سجّل نسبة تثبيط بلغت 96%. كما أظهر المستخلص قدرة عالية على إزالة بيروكسيد الهيدروجين وقوة اختزال لأيونات الحديد، مما يؤكد فعاليته الكبيرة في تقليل الإجهاد التأكسدي وتحيد الجذور الحرة. وتُعزى هذه الفعالية البيولوجية المتميزة إلى وجود مركبات طبيعية فعالة تشمل الفلافونويدات، والأنثوسيانينات، والفينولات، كما تم تأكيدها عبر الفحوصات الكيميائية النباتية. وتخلص الدراسة إلى أن الكركديه (Hibiscus sabdariffa) يُعد مصدرًا طبيعيًا غنيًا بالمركبات الفينولية ومشتقات الأنثوسيانين ذات الخصائص المضادة للأكسدة القوية، مما يجعله مضاد أكسدة طبيعيًا آمنًا ومفيدًا للاستخدام في الصناعات الغذائية والدوائية.
الكلمات المفتاحية: الكركديه (Hibiscus sabdariffa L.)، روزيل، المركبات الفينولية، الأنثوسيانينات، النشاط المضاد للأكسدة، تحليل الكروماتوغرافيا السائلة عالية الأداء (HPLC)، الفحص الكيميائي النباتي، اختبار DPPH، مضادات الأكسدة الطبيعية.
Introduction
Hibiscus sabdariffa L A member of the Malvaceae family, this flower is frequently called roselle. Since its origin in Africa, this species has spread rapidly throughout tropical regions of the planet, primarily in South America , India, Sri Lanka, Indonesia, Malaysia, and Thailand are among the Asian countries, along with Australia. Asia 2’s top producers of roselle are China and Thailand. The calyces of roselle have been used in traditional medicine and can often be swallowed as drinks and snacks. Infusions of roselle calyces have been extensively utilized in the traditional medicine in multiple cultures.)1(. In India, Africa, and Mexico, they are conventionally utilized for their diuretic, cooling, and hypotensive characteristics (2). In Egypt and Sudan, they have been exploited to reduce body temperature. In Thai traditional medicine, roselle has been utilized as an antiviral, hypocholesterolemic, diuretic, mild laxative, and antihypertensive agent(3) . The Thai Herbal Pharmacopoeia 2021 designates the calyces of roselle as a diuretic agent. Further, roselle calyces have been utilized as coloring and flavoring ingredient in food, cosmetics, and medicine (3) n b.
The human body uses antioxidants to protect itself from free radicals and reactive oxygen species (ROS). An excess of reactive oxygen species (ROS) can result in oxidative stress . leading to various pathologies and diseases, such as neurological disorders, rheumatoid arthritis, cancer, and coronary artery bypass surger (4). Natural antioxidants created by dietary sources and plants have garnered much interest. Roselle calyces constitute polyphenolic acids, flavonoids, and anthocyanins, which behave as strong antioxidants.The aqueous extracts of roselle calyx exhibited in vitro antioxidant qualities as assayed by multiple methodologies (5).
Numerous infectious diseases cause medical conditions due to the emergence of strains of microbe and the development of antibiotic resistance. It has generated substantial global public health problems that impose a considerable burden both patient morbidity and financial issues. Also, the administration of antibacterial drugs for bacterial infections is the usual treatment for both human and equine settings. Accordingly, novel antibacterial agents derived from botanical sources for veterinary treatment have garnered much interest in recent years. Methanol extracts of Roselle calyx exhibit an antibacterial effect toward bacteria of all kinds (9). The plant contains multiple bioactive phytochemicals, comprising flavonoids (flavanols, flavanones, and anthocyanins), phenolic acids (hydroxybenzoic and hydroxycinnamic acids), and chemicals responsible for the red color of the caly.(7). A number of factors have been documented that may pH, temperature, enzymes, light, and oxygen all affect the stability of anthocyanins, particularly during the extraction process for red cabbage extract (8). Green extraction pertains to improvements of extraction methodologies that utilize reduced energy consumption and recyclable solvents, while ensuring the safety and quality of extract products (9). Other methodologies, Extractions that are aided by ultrasound, homogenizers, and microwaves are examples of treatments that meet environmentally sensitive standards. This study applies green extraction and partial purification to yield roselle extracts and fractions with better yields , high active component concentrations, and significant biological activity.
MATERIALS AND METHODS
Obtaining Samples, Local markets in Najaf were the source of the dried hibiscus blooms. Healthy flowers that showed no indications of damage or flaws were chosen. Cleaning the Sample, To get rid of dust and other contaminants, the flowers were cleansed and rinsed under running water. After that Following the purification process, they received permission to air dry at ambient temperature. Drying the Sample after disinfecting, the samples were either completely dried in a well-ventilated , dark environment for seven to ten days, or they were dried in a drying oven set to a low temperature of 40 to 45°C to retain the active compounds. Pulverize a Sample An electric grinder was used to create a fine powder from the dried flowers. The powder was kept until it was needed in sterile, tightly sealed containers in a dark, dry location .
Ultrasonic Extraction of Phenolic Compounds:
20 g of the plant was thoroughly chopped, put on an electric vibrator for three hours to remove fat, and then the chloroform layer was removed in order to extract the phenolic chemicals from the homogenized plant. To make sure that no chloroform residues remained, the sample was then dried at 50 °C. Ten grams of the dried sample were then extracted using a solvent made of ethanol and water (70/30). For an hour at room temperature, an Ultrasonic Bath (USA) was utilized to extract The extraction yield was measured using 5 mL of the liquid extract after screening. In Slovenia, A spinning evaporator was utilized for removing the solvent, next heating the material under vacuum till reaching an even density at a temperature of 40°C
For defense against oxidative damage, the dried extracts have been preserved in glass vials at forty degrees Celsius until analysis. Phase inversion . Each unique phenolic compound’s amount was ascertained using HPLC analysis . Sykam A UV detector and a -C18-OSD column (25 cm, 4.6 mm) were included in the HPLC capillary system . Thirty degrees Celsius was the column’s temperature. Gradient elation requires the availability of A (methanol) and B (1% formic acid in water (v/v ) . The flow was 1.0 mL/min, and the ratio of B was 40% during the first 6 minutes and 50% during the next 6-15 minutes. 100 microliters of standards and 100 microliters of samples were automatically introduced using the autosampler. We measured the spectra at 280 nm (10) . To determine the phenolic compound concentration, the following formula was utilized.:
Concentration of the compound ( mg/g) =
Standard concentration * Sam ple peak a rea × Dilution factor
Standard peak are a Sample weight (g)
Standard concentration Concentration of the known standard ( mg ̸ g or µg ̸ mL )
Sample peak area → Peak area of the compound in the sample as determined by HPLC. Standard peak area → Peak area of the standard compound.
Dilution factor → The variety of times the sample was thoroughly diluted ( e.g. , 1 ,10 , etc.)
Sample weight (g) → The dry weight of plant material used for extraction
DPPH analysis Antioxidant activity is measured using 2,2-diphenyl-1-picrylhydrazyl . It was evaluated how well the plant extract interacted with and inhibited free radicals
Preparing a sample to identify anthocyanin chemicals
About 1.0 g of this substance has been put into 20 mL vials. After that, two milliliters of Dimethyl Sulfoxide (DMSO) were added, and the vials were vortexed for a minute .
The vials were allowed to sit in the dark for ten minutes, followed by the addition of eight milliliters of methanol that contained 0.5 mg/mL tert-butylhydroquinone and 2% HCl . The containers were mixed with a vortex mixer for five minutes. Regarding HPLC analysis After going through a 0.2 mm syringe filter, the supernatant solutions were then poured into a secure glass container.
Analyzing anthocyanin chemicals using the HPLC technique
Germany’s SYKAM lab model Anthocyanin measurement and analysis were the goals There is an isocratic flow of a 95/5 (v/v) mixture of water (pH 7.0) and 2% formic acid in the mobile phase . at a rate of 0.8 grams per month. C18-ODS (25 cm x 4.6 mm) was the column used, and UV-Vis (520 nm) was the detector used.
To determine the concentration of anthocyanin chemical components, the second formula is utilized:
Concentration of the compound ( mg/g) =
Standard concentration * Sam ple peak a rea ×Dilution factor
Standard peak are a Sample weight (g)
Standard concentration Concentration of the known standard ( mg ̸ g or µg ̸ mL )
Antioxidant test
DPPH assay :
DPPH has been solubilized in 100 mL of methanol. The DPPH 0.04 g concentration is 400 µg/ml . For creating 100 milliliters of ethanol and distilled water were combined with 0.5 grams of vitamin C and the standard solution.. The default solution quantity was 5000 ppm, and the subsequent dilution treatment provided the following concentrations: 30, 60, 120, 250, and 500 ppm amounts of the antioxidant vitamin C and the sample . The mixture was thoroughly combined and allowed to settle for half an hour at a suitable temperature The absorbance at 517 nm was then measured using a Shimadzu UV-VIS spectrophotometer The inhibitory capacity (IC50) of the sample, Using a monotonic dose-inhibition curve . DPPH free radical concentration required to neutralize 50% of the radical was computed. The reaction mixture’s declining absorbance indicated an increase in free radical activity. The levels of DPPH scavenging activity was determined using the following expression: A0 – A1 / A0 × 100 is the formula for the DPPH scavenging effect (%) or percent inhibition .
A0 represents the absorbance of the blank, and A1 represents the absorbance when the test product is present .
The Peroxide Hydrogen Hunting Potential:
The ability of the supplement to eliminate peroxide from the air A phosphate buffer with a pH of 7.4 has been used to create a 40 milliliter hydrogen peroxide solution. In purified water, There was a hydrogen peroxide solution (0.6 mL, 40 mM) combined with extracts (100 µg/mL). . Ten minutes later, the reactivity of hydrogen-containing peroxide was assessed at 230 nm using a blank solution made of phosphate buffer that had been stripped of hydrogen peroxide.
Using the formula below. Both the extract and standard compounds’ percentage of hydrogen peroxide scavenging was calculated.: % Scavenged [H2O2] = [(AC – AS)/AC] x 100, where AS represents the absorbance in the presence of the sample extracts and AC represents the absorbance of the control.
Capacity Decrease Evaluation:
Efficiency of This technique was used to evaluate the extracts’ capacity to lower ferric ions (Fe+3). After 800 µl of extract was dissolved in 400 µl of phosphate buffer (0.2 M, pH 6.6) and 800 µl of a solution of 1% potassium ferricyanide [K3Fe(CN)6], For 20 minutes at a time, the mixture was incubated at 50 °C. After that, around 800 µl (10%) of TCA was added, and it underwent a 10-minute processing time at 3000 twists per minute. throughout the final stage The absorbance at 700 nm was measured after the precipitate was combined with 400 µl of distilled water and 80 µl of 0.1% FeCl3. The enhanced absorbance of the reaction mixture indicated better reducing power. Micrograms of ascorbic acid equivalent per grain sample are used to express the results.
Phytochemical investigation
Including both qualitative and quantitative elements, the precipitate was 400 µ. Proven phytochemical techniques revealed the existence of particular phytoconstituents. Analyzing anthocyanins, flavonoids, and antioxidants using phytochemistry
Following the strategies outlined in the literature, the precipitate was blended 400 µ. (11, 12). High-performance liquid chromatography and spectrophotometric methods have been used to conduct quantitative and qualitative research on phenolic, flavonoid, and anthocyanin molecules.
Assessment of all phenolic compounds
The procedure developed by Folin-Ciocalteu was used for assessing the freeze -dried extract’s total phenolic content (TPC) (13). The components listed below have been combined: 0.8 milliliters of purified water and 0.5 milliliters of Folin-Choclate’s reagent (diluted 1:9 with water), 0.2 mL of the sample extract (1 mg of freeze-dried extract diluted in 1 mL of methanol) and 1.5 mL of sodium carbonate (17%, w/v). The tubes were left to incubate at room temperature in the dark for 40 minutes prior to measuring absorbance at 765 nm.
Statistical Testing
Version 7.5 of the Statistica SPSS data analysis program was used to investigate the data. Means were separated using the Newman Keuls range test at P < 0.05 and analysis of variance (ANOVA) was employed. The information is a three-replica median. To determine whether the antioxidants’ hindering percentages differ significantly (P<0.05), the Kruskal-Wallis test is used.
Result and discussion
Chemical Analysis of Chemical Compounds in Hibiscus Extract Using HPLC
High-performance liquid chromatography (HPLC) has been employed to precisely recognize and describe the active phenolic components in hibiscus extract through chemical analysis. Descriptive identification, which compares the standard compound’s retention time to the sample’s earliest appearance, was accomplished by injecting numerous well-known standard compounds (standards). The compounds were grouped together. The results were used to identify seven primary phenolic compounds in the extract, as well as additional compounds that appeared in the chromatogram during the brief retention periods. We determined the relative concentration of each compound using the curves of the injected standards. One of the substances that was present in considerable quantities in the extract was chlorogenic acid, which had the longest retention period of all of the chemicals discovered, as seen in Figure(1).
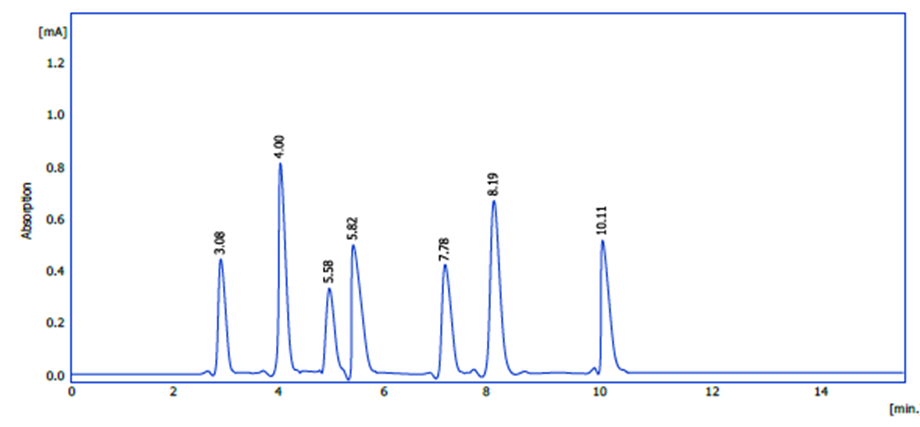
figure 1: HPLC profile of phenolic chemicals from hibiscus petal extract.
These molecules are present because to the extract’s multiple and abundant phenolic component composition, which stimulates biological activity, notably antioxidant activity. Following that, an estimate of their amounts was made and is presented in Table (1). At 145.6 parts per million, rutin had the highest concentration, while chlorogenic acid had the lowest.
Table (1): Concentrations of phenolic compounds in hibiscus flower extract using HPLC analysis.
|
Name |
Con. ( ppm ) |
|
|
1 |
Ferulic acid |
88.0 |
|
2 |
Caffeic acid |
74.2 |
|
3 |
44.8 |
|
|
4 |
Rosemaric |
98.7 |
|
5 |
145.6 |
|
|
6 |
Qurcetine |
123.5 |
|
7 |
Kaempferol |
91.5 |
Determination of anthocyanin compounds by HPLC method
HPLC analysis of the hibiscus flower extract was performed using a multi-wavelength dichroic detector (DAD). The chromatogram showed four main peaks at retention times of 4.50, 5.70, 6.00, and 8.20 min (Figure 2). The highest recorded signal was observed at 5.70 min, while the lowest peak was at 4.50 min. This variation in peak intensities is primarily explained by the variability in the concentrations of the compounds in the extract and the difference in the molar absorbance of each compound at the measured wavelength, in addition to the possibility of partial overlap between two compounds with close retention times (5.70 and 6.00 min). Retention times reflect differences in the structural polarity of the compounds, as less polar compounds tend to retain for longer periods on the reversed-phase column and appear at longer retention times (e.g., the peak at 8.20 min). To obtain accurate quantitative and qualitative determinations, peak areas were calculated, and the results were compared with calibration curves of standard anthocyanin standards.
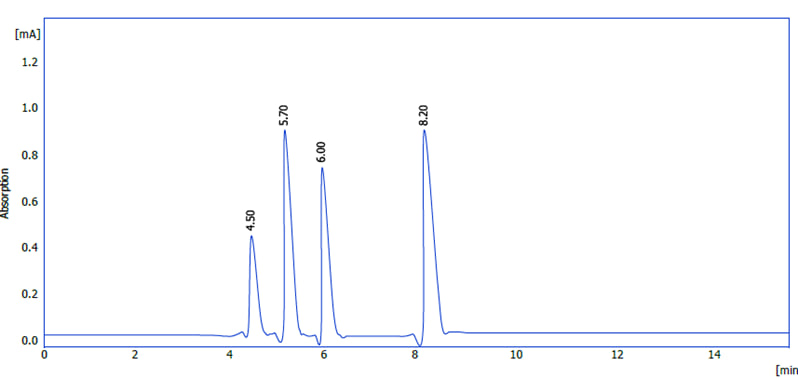
Figure (2) : HPLC chromatogram of Hibiscus sabdariffa extract showing main anthocyanin compounds .
Reverse phase HPLC was used to separate Hibiscus sabdariffa’s four different anthocyanins (Figure 2). Peak assignments can be determined by comparing identical HPLC retention durations and UV-vis spectra with known anthocyanins from a reference library of previously purified compounds. According to several publications, anthocyanins that have been derived from Roselle petals have pharmacological characteristics (14, 15, 11, 12, 16). Chromatograms indicated that delphinidin (peak 1) and cyanidin 3-O-sambubioside (peak 2) are the primary anthocyanins. Delphinidin (peak 4) and cyanidin 3-O-glucoside (peak 3) had absorbances four times higher than these two anthocyanins. Many authors have observed that the petals of Roselle contain anthocyanins (17, 18). Once identified, the amounts of each anthocyanin molecule were calculated using the formula and contrasted with those of similar substances. As indicated in table (2) Table (2): Concentrations of anthocyanin compounds in hibiscus flower extract using HPLC analysis.
|
No ( ppm ) |
Cyanidin |
Malvidin |
Delphinidin |
|
Extract |
78.7 |
91.4 |
71.9 |
Evaluation of antioxidant activity using DPPH analysis
A DPPH assay was used to assess the antioxidant activity of hibiscus extract and standard vitamin C. A variety of concentrations (30, 60, 120, 250, 500, and IC50) ppm were used to gauge the rate of free radical inhibition. The extract recorded the maximum inhibition rate of 96.00 at an IC50 concentration, while the results in Figure (3) demonstrated the highest inhibition rate of vitamin C (67.00) at a concentration of 500 ppm. The findings show that, at the same doses, hibiscus extract has greater antioxidant activity than vitamin C. This is because phenolic chemicals, which are renowned for their strong capacity to suppress free radicals, include anthocyanins, caffeic acid, and chlorogenic acid
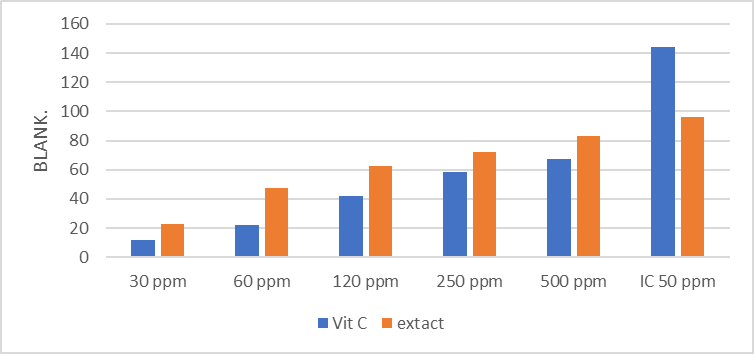
Figure (3): Impact of antioxidant activity on ascorbic acid and hibiscus flower extract DPPH analysis. The conclusion is the standard deviation of the five individual concentrations that were studied.
Effect of extract concentration on the ability to reduce hydrogen peroxide:
The percentage increased from 44.08% at a concentration of 1% to 74.59% at a concentration of 2.5%, showing that the plant extract’s ability to lower hydrogen peroxide gradually improves as extract concentration rises, as seen in Figure (4). This behavior demonstrates the extract’s efficacy in lowering oxidative stress and shows a direct correlation between concentration and antioxidant capacity. Flavonoids and phenolic chemicals found in the extract can donate protons or electrons to lower H2O2 and turn it into H2O, lessening the damage it causes to cells. As a secondary free radical, hydrogen peroxide undergoes Fenton reactions in the body to produce extremely harmful hydroxyl radicals (OH−). As a result, this activity shows antioxidant capacity. The hibiscus extract’s potent capacity to block H2O2 is demonstrated by the maximum decrease rate at a concentration of 2.5%, confirming that the extract’s active ingredients become increasingly capable of interacting with free radicals as their concentration rises. According to their findings, antioxidant activity improved as plant extract concentrations rose.
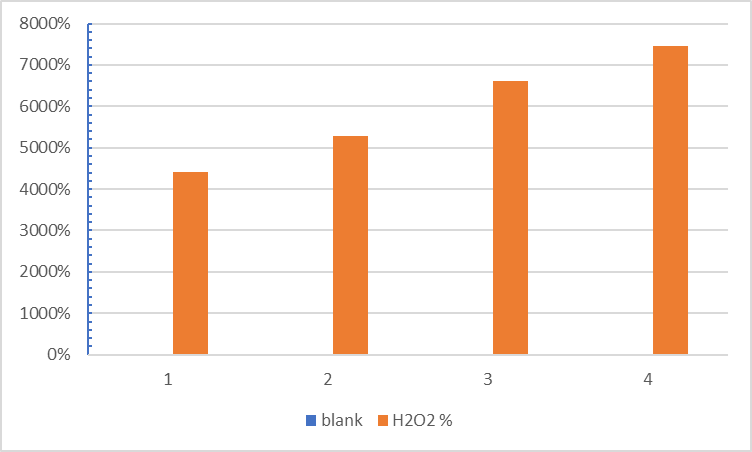
Figure (4) : Effect of hibiscus flower extract concentration on H2O2 scavenging capacity at three concentrations.
Effect of extract concentration on reducing capacity:
The findings in Figure (5) demonstrated that the plant extract’s antioxidant activity and Fe3 to Fe2 decrease were significantly greater than those of vitamin C at all doses. This suggests that the extract has flavonoids and phenolic chemicals that are quite efficient.able to change ferrous ions (Fe3) into Fe2 and donate electrons. The reducing power rose from 40.23% to 72.65% as the extract concentration rose from 2% to 6%, suggesting a direct correlation between concentration and activity (2). While not quite as much as the extract, in comparison to vitamin C’s reducing power, the reducing power rose from 18.08% to 41.09% at the same concentration. The significant reducing power of the hibiscus extract indicates that it contains a large number of compounds that can donate electrons to halt the oxidative chain reaction. In a study by (19), Ethanol, acetone, and chloroform were the three solvents used to extract the leaves of Epipremnum aureum . Thin layer chromatography and preliminary methods of detection were employed to look for phytochemicals in the plant extract. Tests have been conducted on the crude plant extract’s antioxidant activity at different levels, reducing power activity and activating DPPH scavenging activity. During an initial qualitative chemical screening, numerous extracts contained steroids, terpenoids, alkaloids, flavonoids, tannins, and saponins.
Effectiveness against free radicals was demonstrated by all three extracts. The ethanol extract had greater antioxidant activity than the acetone and chloroform.
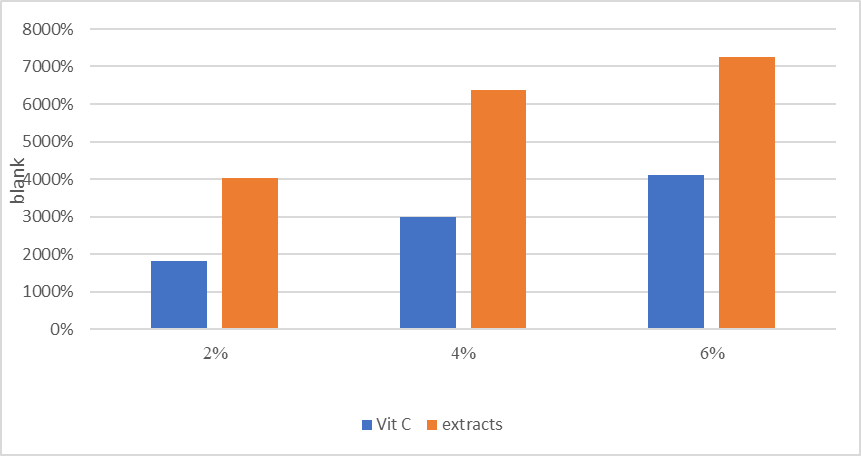
Figure (5) : The impact of three dosage levels of hibiscus flower extract on ascorbic acid and diminished capacity
Phytochemical screening
Numerous researchers carried out evaluations of plants for medicinal value through preliminary phytochemical analysis (20). Phytochemical screening investigations serve as essential for identifying new sources of therapeutically and industrially useful chemicals, hence increasing the use of natural assets that exist. Numerous therapeutic plants have been chemical examination by multiple experts (21, 22, 23). The identification of plant constituents, such as petals, resulting in the largest number of secondary metabolites is the vital preparatory phase of the investigation . Multiple phytochemicals have been separated from the petals using specific methods, with their presence (+) or lack (-) stated in Table 3. Studies have shown that the main phytochemical classes exhibiting biological activity in The compounds found in rosella petals include tannins, steroids, sterols, anthocyanins, flavonoids, alkaloids, and saponins. Variations in genetics, soil type, and the extractive solvent used could have been the cause of some of the variations in the content of the aqueous extract of Roselle petals, which matched previous studies. (24, 20, 23). Anthocyanins are recognized for their cardioprotective, hypocholesterolemia, antioxidative, and hepatoprotective properties (25, 26). They exhibit antioxidant properties and impede the oxidation of low-density lipoprotein (LDL) (27). Phenolic pollutants are attracting significant scrutiny due to the possibility of therapeutic uses. In recent years, multiple laboratory experiments have established the biological and pharmacological implications of organic chemicals. The petals of Roselle contain helpful phytoconstituents that may assist in getting rid for several ailments in the future.
Table (3): displays the concentrations of anthocyanins, flavonoids, and phenols in flower extracts from Hibiscus sabdariffa.
|
N |
Test |
Aqueous extract |
|
1 |
Phenols |
+ |
|
2 |
Saponins |
+ |
|
3 |
Tannins |
+ |
|
4 |
Alkaloids |
+ |
|
5 |
Terpenoids |
– |
|
6 |
Anthocyanins |
+ |
|
7 |
Steroids and sterols |
– |
|
8 |
Flavonoids |
+ |
|
9 |
Quinones |
– |
(+) Present; (-) Absent
Discussion
illustrates the variations in antioxidant activity between ascorbic acid and the extract. The capacity of rosella petals to scavenge DPPH radicals demonstrated a dose-dependent relationship (Figure 3). A significant inhibitory effect on the DPPH radical was observed at all extract concentrations. Importantly, all extract doses showed higher levels of antioxidant activity than ascorbic acid. The results of multiple tested concentrations are used to determine the amount of the radical-scavenging impact that must be blocked at 50% (IC50). The IC50 value decreases, indicating increasing scavenging action. Ascorbic acid, the reference antioxidant used in this study, has an IC50 value of 96.0 mg/ml, whereas the Roselle extracts was 96.0 mg/ml., was 144.2 mg/ml. Findings indicated an important amount of phytochemicals with antioxidant properties found in Roselle petal extracts. Free radicals are recognized to have the ability to damage biomolecules, and their production is excessive, is stimulated by oxidative stress. (28). Roselle may act as a free radical scavenger or decrease the production of free radicals, according to antioxidant research. Our extract from H. Sabdariffa showed results that were comparable to those previously published by (15). Studies on antioxidants show that Roselle may either act as a free radical scavenger or limit the production of free radicals. The results from our H. Sabdariffa extract were comparable to those previously published by (29). Roselle petal extracts’ higher antioxidant activity is attributed to the presence of polyphenolic components such as anthocyanins, flavonoids, and phenolic acids. According to research on antioxidants, Roselle may either act as a free radical scavenger or inhibit the production of free radicals.
Conclusion and recommendations
The results of our H. Sabdariffa extract were comparable to those previously published by. The primary source of polyphenolic compounds’ antioxidant capacity is their redox characteristics, which are critical for capturing and eliminating harmful free radicals, deactivating singlet and triplet oxygen, and breaking down peroxide. Plants typically include polyphenolic chemicals such flavonoids, phenolic acids, and anthocyanins, which have been demonstrated to have a range of biological effects, among these, antioxidant activity is the most important (23, 30). All samples showed a decrease in antioxidant activity as extract concentration increased.
Acknowledgments
The authors thank the University of Kufa, College of Science, Department of Ecology Science, for providing facilities and academic support. Appreciation is extended to colleagues for their assistance during field and laboratory work.
References
- Islam, A. K. M. A., Osman, M. B., Mohamad, M. B., & Islam, A. K. M. M. (2021). Vegetable Mesta (Hibiscus sabdariffa L. var sabdariffa): A potential industrial crop for southeast Asia. In S. M. Sapuan, R. Nadlene, A. M. Radzi, & R. A. Ilyas (Eds.), Roselle (pp. 25–42).
- Ilyas, R. A., Sapuan, S. M., Kirubaanand, W., Zahfiq, Z. M., Atikah, M. S. N., Ibrahim, R., Radzi, A. M., Nadlene, R., Asyraf, M. R. M., Hazrol, M. D., Sherwani, S. F. K., Harussani, M. M., Tarique, J., Nazrin, A., & Syafiq, R. (2021). Roselle: Production, product development, and composites. In S. M. Sapuan, R. Nadlene, A. M. Radzi, & R. A. Ilyas (Eds.), Roselle (pp. 1–23).
- Chumsri, P., Sirichote, A., & Itharat, A. (2008). Studies on the optimum conditions for the extraction and concentration of roselle (Hibiscus sabdariffa Linn.) extract. Songklanakarin Journal of Science & Technology, 30(supp.1), 133–139.
- Pham-Huy, L. A., He, H., & Pham-Huy, C. (2008). Free radicals, antioxidants in disease and health. International Journal of Biomedical Science, 4(2), 89–96.
- Mohamed, B. B. (2021). Roselle (Hibiscus sabdariffa L.) in Sudan: Production and uses. In S. M. Sapuan, R. Nadlene, A. M. Radzi, & R. A. Ilyas (Eds.), Roselle (pp. 121–127).
- Abdallah, E. M. (2016). Antibacterial efficiency of the Sudanese roselle (Hibiscus sabdariffa L.), a famous beverage from Sudanese folk medicine. Journal of Intercultural Ethnopharmacology, 5(2), 186–190.
- Pinela, J., Prieto, M., Pereira, E., Jabeur, I., Barreiro, M. F., Barros, L., & Ferreira, I. C. (2019). Optimization of heat-and ultrasound-assisted extraction of anthocyanins from Hibiscus sabdariffa calyces for natural food colorants. Food Chemistry, 275, 309–321.
- Ghareaghajlou, N., Hallaj-Nezhadi, S., & Ghasempour, Z. (2021). Red cabbage anthocyanins: Stability, extraction, biological activities and applications in food systems. Food Chemistry, 365, 130482.
- Thajeel, Z. H., Guda, M. A., Mutlag, N. H., & Jabbar, B. S. (2020). Investigation the effect of irrigation with magnetized saline water to improve the vegetative growth and the anatomical characteristics of the tomato plant (Lycopersicon esculentum L.). International Journal of Agricultural and Statistical Sciences, 16(1), 1551-1557.
- Qazmooz, H. A., Guda, M. A., Algburi, J. B., Al-Zurfi, S. K., & Al-Graiti, T. A. (2020). DETERMINATION OF HEAVY METAL IN SAMPLES OF TIRMANIA NIVEA FUNGI IN DIFFERENT SOILS. Plant Archives, 20(2), 313-317. http://www.plantarchives.org/SPL%20ISSUE%2020-2/50__313-317.pdf
- Mutlag, N. H., Juda, M. A., Hussein, M. L., & Hassan, H. N. (2019, September). Assessment of Bombax ceiba Leaves Extract and Pleurotus ostreatus Fungus Filtrate on treatment of some Isolated Dermatophytic Fungi. In Journal of Physics: Conference Series (Vol. 1294, No. 7, p. 072022). IOP Publishing.
- Hakeem, G. L., Guda, M. A., Alabassi, M. M., Altamimi, A. J., & Alhadrawi, H. A. (2020, June). Use of wild plant species as indicator of some heavy metals in the soil of General Company for tire industry in Najaf Governorate. In IOP Conference Series: Materials Science and Engineering (Vol. 870, No. 1, p. 012099). IOP Publishing. https://iopscience.iop.org/article/10.1088/1757-899X/870/1/012099/pdf
- Guda, M., Taher, M., & Almayahi, B. (2019). Anatomical characteristics of vascular bundles associated with heat tolerance in Phragmites australis. Analele Universitatii din Oradea, Fascicula Biologie, 26(2), 136-139.
- Guda, M. A., Yoness, A. S., Mohammed, S. J., & Alasedi, K. K. (2021). Enzymatic and anatomical responses of wheat (Triticum aestivum L.) Cultivar IPA 99 to irrigation with magnetized water. Iranian Journal of Ichthyology, 8, 280-289.
- Guda, M. A., Obaid, J. K., Alkurdi, H. J., Hakeem, G. L., Salih, H. M., & Isawi, H. F. A. (2020, June). The role of street and park trees to CO2 removal and improving air quality and climate in urban areas in Najaf (Iraq) Type the title of your paper here. In IOP Conference Series: Materials Science and Engineering (Vol. 870, No. 1, p. 012087). IOP Publishing. https://iopscience.iop.org/article/10.1088/1757-899X/870/1/012087/pdf
- Guda, M. A., OBAID, J. K. , ALASEDI, k.k., ALDUHAIDAHAWI, F. J.(2020). Comparison of Vascular plant leaves in the ability to collect Atmospheric heavy metals in urban areas, International Journal of Pharmaceutical Research, 13(1),47-52. DOI: 10.31838/ijpr/2021.13.01.014
- Guda, M. A., Nasir, A. S., Younus, A. S., & Altamimi, A. J. (2018). Antioxidant enzyme responses of Juncus Aschers.(Et Buch.) Adams to some of environmental stresses and use it as indicators. EXECUTIVE EDITOR, 9(12), 1102.
- Guda, M. A., Mutlag, N. H., & Tsear, A. A. (2020, December). The use of Atriplex nummularia plant as the hyperaccumulators of silver. In AIP Conference Proceedings (Vol. 2290, No. 1, p. 020041). AIP Publishing LLC.; https://doi.org/10.1063/5.0027565
- Guda, M. A., Mutlag, N. H., & Alasedi, K. K. (2018). The potential use of Atriplex nummularia plant as contamination indicators of heavy metal in different soils. Plant Archives, 18(2), 2372-2378.
- Guda, M. A., Merza, T., & Almayahi, B. (2016). Response of non-enzymatic antioxidants to phragmites Australis (Cav.) Trin. Ex. Steudel Plants of the Environmental Stresses in Baher Alnajaf, Iraq. Plant Cell Biotech Molec Biol, 17, 140-148.
- Guda, M. A., Hakeem, J. I., Alabassi, M. M., & Almayahi, B. A. (2017). Effects of Environmental Stress on Nutrients of Typha domingensis Pers. Plant in Najaf, Iraq. Annual Research & Review in Biology, 1-6. http://journalarrb.com/index.php/ARRB/article/view/26235
- Guda, M. A., Hakeem, J. I., Alabassi, M. M., & Almayahi, B. A. (2020). Phytoremediation of Some Heavy Metals in The Soil of General Company for Tire Industry in Najaf Governorate by Wild Plant Species. Indian Journal of Environmental Protection, 40(7), 758-763.
- Guda, M. A., Alkaabiº, Z. S., Albayati, F. S., Alduhaidahawi, F. J., & Almayahi, B. A. Anatomical And Sclerophilic Traits Variation ln Two Varieties Of Olive Plants (Olea europaea L) Growing Under Climate Changes ln Various.season of the year. Indian Journal of Environmental protection,Vol.40,No.1Junuary 20,77-83.
- Guda, M. A., & Obaid, J. K. (2019). The Spatial Variation Study of Spread the Lupus Erythematous Disease in Najaf and Babylon Governorates. Indian Journal of Forensic Medicine & Toxicology, 13(2).
- Al-Khafaji, B. A., Guda, M. A., Al-Edhari, A. H., & Altamimi, A. J. (2019). Taxonomic Study for the Genus Bupleurum L.(Apiaceae) in Iraq using chloroplast gene RPL16. Indian Journal of Public Health, 10(01).
- Al-Edhari, A. H., Shayma’a, M., Sardar, A. S., Guda, M. A., & Almayahi, B. A. (2018). MORPHOLOGICAL STUDY FOR VALERIANELLA KOTSCHYI BOISS. ANDV. MURICATA (STEV.) BAXT.(VALERIANACEAE) IN KURDISTAN, IRAQ. Plant Archives, 18(2), 2417-2424.
- Alduhaidahawi, F. J., Guda, M. A., Al-Graiti, T. A., Abdul-Hussein, A., Al Regawi, S. M., & Al-Edhari, A. H. (2009). Environmental Analysis for Spreading Areas of Scleroderma Disease in Babylon Governorate.
- Alabassi, M. M., Al Naqeeb, N. A., Guda, M. A., & Jabbar, B. S. (2020). The study of drinking water pollution by studying some chemical and biological characteristics in Najaf province, Iraq. EurAsian Journal of BioSciences, 14(2), 7159-7164.

