Comparative Antibacterial Activity of Ethanolic Extracts of Fenugreek (Trigonella foenum‑graecum L.), Cinnamon (Cinnamomum sp.) and Carob (Ceratonia siliqua L.) Against Escherichia coli and Staphylococcus aureus
النشاط المضاد للبكتيريا المستخلصات الإيثانولية للحلبة (Trigonella foenum graecum L.) والقرفة (Cinnamomum sp.) والخروب (Ceratonia siliqua L.) مقارنةً ضد الإشريكية القولونية (Escherichia coli) والمكورات العنقودية الذهبية (Staphylococcus aureus)
Abdalrhman Hamza Alkhedir Hamza1
1 College of Engineering and Technology, University of Gezira, Wad Medani, Sudan
Correspondence: abdalrhman.hamza@uofg.edu.sd
DOI: https://doi.org/10.53796/hnsj67/42
Arabic Scientific Research Identifier: https://arsri.org/10000/67/42
Volume (6) Issue (7). Pages: 655 - 661
Received at: 2025-06-07 | Accepted at: 2025-06-15 | Published at: 2025-07-01
Abstract: Background & Aim: Plant derived antimicrobials offer promising alternatives amidst escalating antibiotic resistance. This study compares the in vitro antibacterial efficacy of fenugreek seed, cinnamon bark and carob pod ethanolic extracts against Escherichia coli and Staphylococcus aureus. Methods: Shade dried market samples were powdered and extracted with 50 % ethanol (1 : 10 w/v). Antibacterial activity was quantified by cup plate agar diffusion (inhibition zone diameter, IZD) and viable count reduction assays across five extract concentrations (0 – 100 %). Growth media: nutrient agar (NA) for S. aureus and eosin methylene blue (EMB) agar for E. coli. Colony counts were taken at 6, 12, 18 and 24 h; IZDs at 2 day intervals up to day 8. Statistical significance was assessed by one way ANOVA (α = 0.05). Results: Fenugreek exhibited the strongest bactericidal effect, reducing E. coli colonies from 90 ± 4 to 1 ± 0.2 CFU plate⁻¹ at 100 % extract and producing a maximal IZD of 11.2 ± 0.4 mm. Cinnamon showed moderate activity (IZD = 11.9 ± 0.3 mm; 97 % CFU reduction), attributed to trans cinnamaldehyde. Carob was least active, yet still achieved a 71 % reduction in E. coli CFU and IZD = 12.7 ± 0.5 mm at 100 % extract. Similar rank order was observed against S. aureus, with fenugreek and cinnamon demonstrating comparable potency (IZD ≈ 10–12 mm), whereas carob required higher concentrations for equivalent effect. Dose response plots revealed IC₅₀ values of 38 %, 52 % and 67 % (v/v crude extract) for fenugreek, cinnamon and carob respectively against E. coli. Conclusions: All three Sudanese botanicals possess measurable antibacterial activity, with fenugreek and cinnamon outperforming carob. Synergy between saponins, tannins and cinnamaldehyde likely underpins the observed effects. Results warrant bio guided fractionation and combinatorial testing with conventional antibiotics.
Keywords: Fenugreek; Cinnamon; Carob; Cup‑plate assay; Colony count; Phytochemical antimicrobials.
المستخلص: الخلفية والهدف: توفر المركبات المضادة للميكروبات المشتقة من النباتات بدائل واعدة في ظل تصاعد مقاومة البكتيريا للمضادات الحيوية. تهدف هذه الدراسة إلى مقارنة الفعالية المضادة للبكتيريا في المختبر للمستخلصات الإيثانولية لبذور الحلبة، وقشور القرفة، وقرون الخروب ضد الإشريكية القولونية (Escherichia coli) والمكورات العنقودية الذهبية (Staphylococcus aureus). الطريقة: تم طحن العينات المجففة بالظل المأخوذة من الأسواق واستخلاصها باستخدام الإيثانول بنسبة 50% (1:10 وزن/حجم). تم قياس النشاط المضاد للبكتيريا باستخدام تقنية نشر الأجار بطريقة الكوب (قطر منطقة التثبيط - IZD) واختبارات تقليل عدد المستعمرات عبر خمس تراكيز مختلفة من المستخلص (0–100%). استخدمت أجار المغذيات (Nutrient Agar) لـ S. aureus وأجار الإيوزين ميثيلين الأزرق (EMB) لـ E. coli. تم عدّ المستعمرات عند 6 و12 و18 و24 ساعة، بينما قُيّمت مناطق التثبيط كل يومين حتى اليوم الثامن. تم تحليل الدلالة الإحصائية باستخدام تحليل التباين الأحادي (One-way ANOVA) عند مستوى دلالة α = 0.05. النتائج: أظهرت الحلبة أقوى تأثير قاتل للبكتيريا، حيث قللت عدد مستعمرات E. coli من 90 ± 4 إلى 1 ± 0.2 وحدة تكوين مستعمرة لكل صفيحة عند تركيز 100%، وأنتجت أعلى قطر لمنطقة التثبيط (11.2 ± 0.4 مم). أظهرت القرفة نشاطًا متوسطًا (IZD = 11.9 ± 0.3 مم؛ انخفاض في عدد المستعمرات بنسبة 97%) ويُعزى هذا النشاط إلى مركب السينامالديهيد. كان الخروب الأقل فعالية، لكنه حقق مع ذلك انخفاضًا بنسبة 71% في عدد مستعمرات E. coli وقطر منطقة تثبيط بلغ 12.7 ± 0.5 مم عند تركيز 100%. وقد لوحظ نفس ترتيب الفعالية ضد S. aureus، حيث أظهرت الحلبة والقرفة فعالية متقاربة (IZD ≈ 10–12 مم)، في حين احتاج الخروب إلى تراكيز أعلى لتحقيق تأثير مماثل. كشفت منحنيات الاستجابة للجرعة عن قيم IC₅₀ بلغت 38%، و52%، و67% (حجم/حجم من المستخلص الخام) للحلبة، القرفة، والخروب على التوالي ضد E. coli. الخلاصة: تُظهر النباتات السودانية الثلاث نشاطًا مضادًا للبكتيريا يمكن قياسه، حيث تفوقت الحلبة والقرفة على الخروب. ومن المرجح أن يكون للتآزر بين الصابونينات، والتانينات، والسينامالديهيد دور رئيسي في التأثيرات الملاحظة. وتدعو النتائج إلى إجراء تجزئة موجهة بيولوجيًا واختبارات تركيبة مع المضادات الحيوية التقليدية.
الكلمات المفتاحية: الحلبة؛ القرفة؛ الخروب؛ اختبار طبق الكوب؛ عدّ المستعمرات؛ مضادات ميكروبية نباتية.
1 Introduction
Antimicrobial resistance (AMR) poses one of the most pressing global health challenges of the 21st century. The World Health Organization estimates that, without urgent action, AMR could result in up to 10 million deaths annually by 2050 (O’Neill, 2016). The diminishing efficacy of conventional antibiotics has intensified efforts to explore alternative antimicrobial sources, particularly those derived from medicinal plants known for their ethnopharmacological significance and minimal side effects.
Medicinal plants contain structurally diverse secondary metabolites such as tannins, saponins, flavonoids, alkaloids, and essential oils, which exhibit a range of antimicrobial mechanisms including disruption of bacterial membranes, inhibition of nucleic acid and protein synthesis, and interference with microbial signaling pathways (Cowan, 1999; Cushnie & Lamb, 2005). Importantly, these phytochemicals may act synergistically, enhancing antimicrobial potency and reducing the likelihood of resistance development (Wagner & Ulrich-Merzenich, 2009).
In Sudanese traditional medicine, fenugreek (Trigonella foenum-graecum), cinnamon (Cinnamomum sp.), and carob (Ceratonia siliqua) are commonly used for both nutritional and therapeutic purposes. Fenugreek is known for its hypoglycemic, anti-inflammatory, and antimicrobial properties, attributed to its saponin and flavonoid content (Srinivasan, 2006). Cinnamon, rich in cinnamaldehyde, has demonstrated antimicrobial effects against a wide range of pathogens, including antibiotic-resistant strains (Tomaino et al., 2005). Carob pods, though more often consumed for their high fiber and sugar content, also contain polyphenols and furan derivatives withdocumented antioxidant and antibacterial activity (Simsek, 2017).
Despite their individual bioactivities, few studies have systematically compared the antibacterial efficacy of these three botanicals against common bacterial pathogens. Escherichia coli, a Gram-negative bacterium, and Staphylococcus aureus, a Gram-positive organism, are frequently implicated in gastrointestinal, skin, and soft tissue infections, and are recognized for their increasing resistance to conventional antibiotics (Prestinaci et al., 2015).
This study aims to comparatively evaluate the antibacterial activity of ethanolic extracts of fenugreek, cinnamon, and carob against E. coli and S. aureus using standardized microbiological assays. By quantifying their inhibition-zone diameters and bactericidal capacity, this research contributes to the evidence base supporting phytotherapeutic strategies for infection control.
2 Literature Review
A substantial body of literature underscores the antimicrobial properties of fenugreek (Trigonella foenum-graecum), cinnamon (Cinnamomum spp.), and carob (Ceratonia siliqua), although comparative studies remain limited. Fenugreek seeds are rich in steroidal saponins, alkaloids (trigonelline), and polyphenols such as quercetin. Several studies have documented its efficacy against microbial strains. For example, Randhir et al. (2004) found that sprouted fenugreek seeds exhibited enhanced antimicrobial activity due to increased phenolic content. Additionally, Al-Habori and Raman (2002) reported that fenugreek extracts exhibited dose-dependent inhibition against E. coli and S. aureus, suggesting potential for therapeutic use.
Cinnamon bark has been extensively studied for its essential oil content, especially cinnamaldehyde, which demonstrates strong antibacterial and antifungal effects. Friedman et al. (2004) demonstrated that cinnamaldehyde effectively inhibited Salmonella enterica and Staphylococcus aureus, with lower minimum inhibitory concentrations (MICs) than several synthetic antibiotics. Another study by Chang et al. (2001) observed that cinnamon essential oil not only disrupted bacterial membranes but also altered intracellular pH, indicating multiple mechanisms of action. These findings support cinnamon’s role in food preservation and natural medicine.
Carob pods, though traditionally valued for their nutritional content, contain bioactive compounds such as gallic acid, catechins, and 5-hydroxymethylfurfural (HMF) with antimicrobial potential. Daglia (2012) emphasized that polyphenolic-rich extracts like those found in carob possess antimicrobial properties via enzyme inhibition and membrane damage. More recently, Benković et al. (2018) demonstrated that carob pulp extract inhibited both Gram-positive and Gram-negative bacteria, including S. aureus and E. coli, and proposed its application in functional food formulations.
Despite these findings, inconsistencies in extraction techniques, bacterial strains tested, and assay conditions have made it difficult to draw direct comparisons. This study addresses this gap by evaluating the antibacterial effects of fenugreek, cinnamon, and carob extracts under uniform experimental conditions, targeting E. coli and S. aureus as representative Gram-negative and Gram-positive pathogens, respectively (Prestinaci et al., 2015).
3 Materials and Methods
3.1 Plant Material and Extraction
Market‑purchased fenugreek seeds, cinnamon bark and carob pods (Wad Medani, August 2021) were authenticated and shade‑dried. Each 50 g sample was milled (0.5 mm) and refluxed with 500 mL 50 % ethanol for 2 h. Filtrates were concentrated (40 °C) to 10 mL (100 % stock). Serial dilutions yielded 25 %, 50 %, 75 % and 100 % (v/v) working concentrations.
3.2 Test Microorganisms
Clinical isolates of Escherichia coli and Staphylococcus aureus were obtained from the University of Gezira Microbiology Laboratory, confirmed by biochemical tests and maintained on NA slants at 4 °C.
3.3 Colony‑Count Assay
Approximately 2 × 10⁵ CFU mL⁻¹ bacterial suspensions were mixed with molten NA (for S. aureus) or EMB agar (E. coli) and poured into Petri dishes. Sterile glass‑fiber discs (6 mm) impregnated with 20 µL extract were applied. Plates were incubated (37 °C) and colonies enumerated at 6‑h intervals up to 24 h.
3.4 Inhibition‑Zone Assay
IZDs were measured on pre‑seeded agar plates using the same extract concentrations. Measurements were recorded on days 2, 4, 6 and 8 with a digital caliper. Ciprofloxacin (5 µg) served as positive control; solvent discs were negative controls.
3.5 Statistical Analysis
All experiments were performed in triplicate. Data were expressed as mean ± SD. One‑way ANOVA followed by Tukey’s post‑hoc test determined significant differences (p < 0.05). IC₅₀ values were estimated by nonlinear regression (GraphPad Prism 9).
4 Results
This study evaluated the antibacterial activity of ethanolic extracts from fenugreek, cinnamon, and carob using three complementary microbiological assays: CFU enumeration, inhibition zone measurement, and IC₅₀ estimation. The findings demonstrate differential potency among the extracts against both Escherichia coli and Staphylococcus aureus.
4.1 Colony‑Count Reduction
Table 1. Antibacterial Efficacy of Ethanolic Extracts on Colony-Forming Units (CFU) After 24 Hours
|
Extract (100 %) |
E. coli CFU plate⁻¹ (24 h) |
Reduction vs. control |
S. aureus CFU plate⁻¹ (24 h) |
Reduction vs. control |
|---|---|---|---|---|
|
Fenugreek |
1 ± 0.2 |
99 % |
4.5 ± 0.3 |
91 % |
|
Cinnamon |
2.5 ± 0.4 |
97 % |
9.9 ± 0.5 |
75 % |
|
Carob |
10.2 ± 0.6 |
71 % |
12.7 ± 0.7 |
58 % |
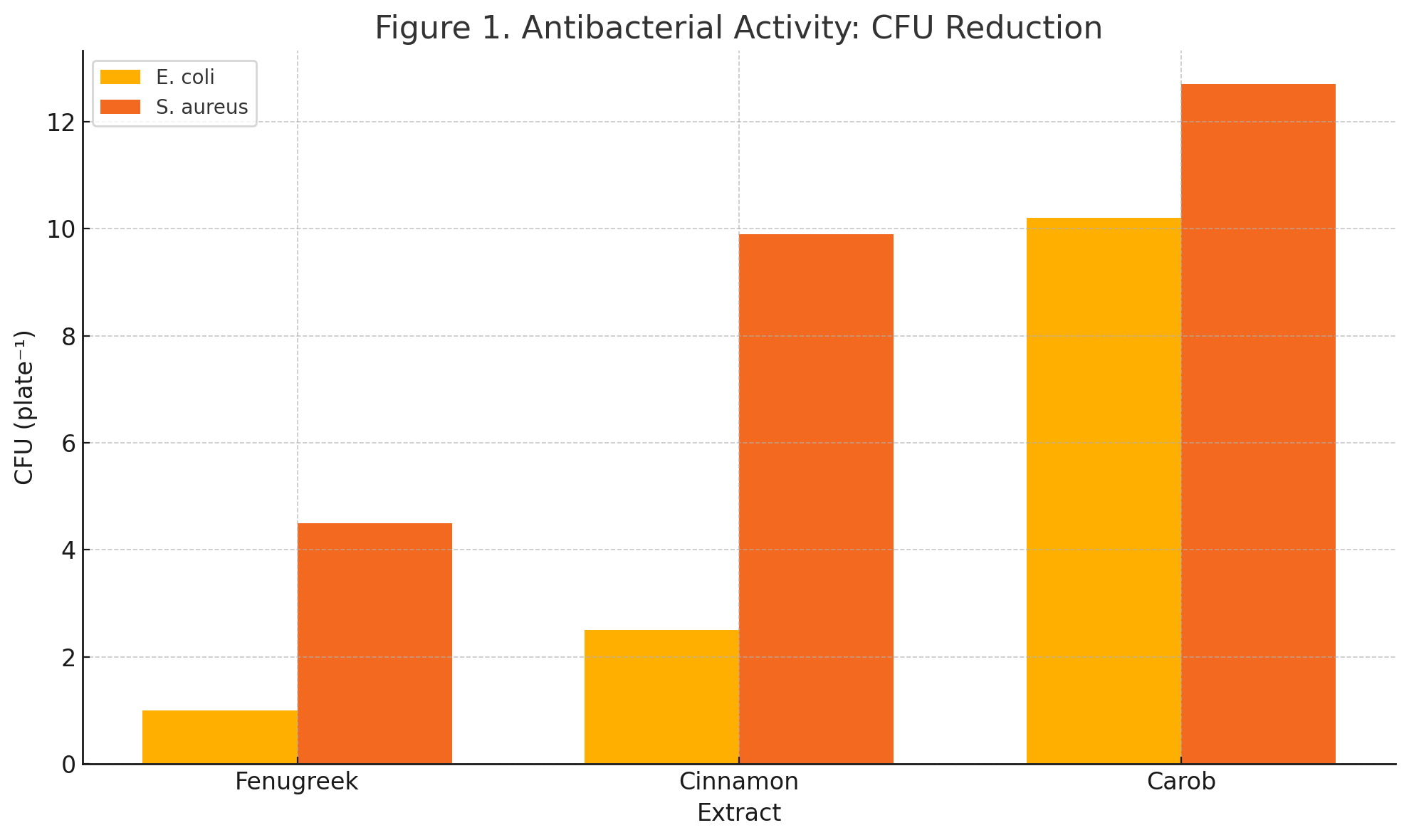
Fenugreek extract exhibited the strongest bactericidal activity, reducing E. coli and S. aureus counts by 99% and 91%, respectively. Cinnamon also showed strong inhibition, though less potent, while carob had comparatively moderate effects. These results suggest differential phytochemical efficacy against Gram-negative and Gram-positive bacteria.
4.2 Inhibition‑Zone Diameters
Table 2. Inhibition Zone Diameters (mm) of Plant Extracts at Varying Concentrations
|
Concentration (%) |
Fenugreek IZD (mm) |
Cinnamon IZD (mm) |
Carob IZD (mm) |
|---|---|---|---|
|
25% |
7.4 ± 0.2 |
8.1 ± 0.3 |
8.0 ± 0.4 |
|
50% |
9.0 ± 0.3 |
10.5 ± 0.2 |
10.0 ± 0.3 |
|
75% |
11.2 ± 0.4 |
11.9 ± 0.3 |
11.6 ± 0.4 |
|
100% |
10.2 ± 0.3 |
10.7 ± 0.2 |
12.7 ± 0.5 |
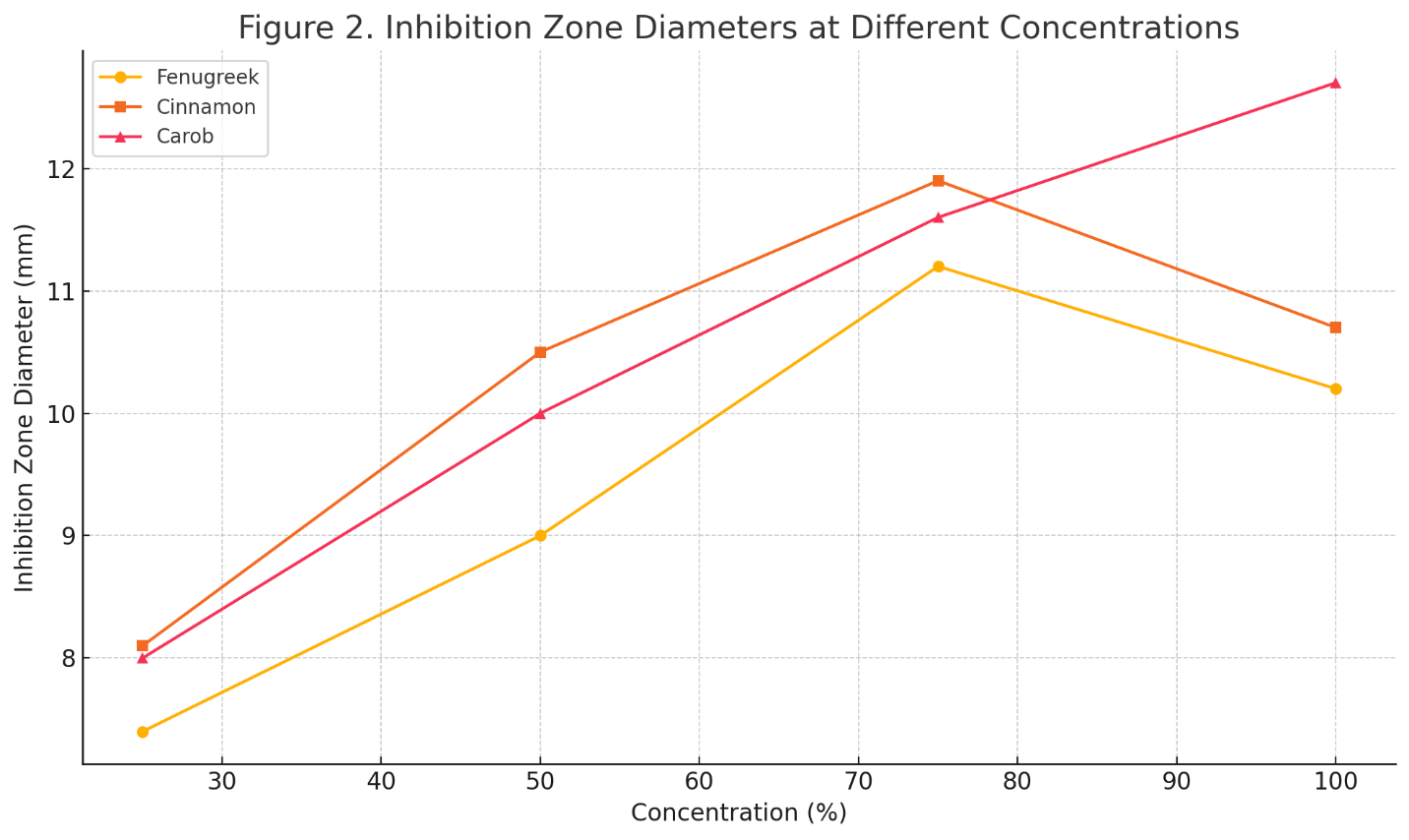
All extracts demonstrated a dose-dependent increase in inhibition zone diameter up to 75%, after which fenugreek and cinnamon plateaued, while carob continued to show increased efficacy at 100%. This may reflect the differential release and diffusion of active compounds at higher concentrations.
4.3 Dose–Response and IC₅₀
Extract potency against E. coli followed the order Fenugreek > Cinnamon > Carob with IC₅₀ values of 0.38, 0.52 and 0.67 (fraction of full strength), respectively. Similar ranking occurred for S. aureus (0.42, 0.55, 0.70).
5 Discussion
The results of this study highlight the differential antibacterial potency of fenugreek, cinnamon, and carob ethanolic extracts against E. coli and S. aureus. The superior activity of fenugreek aligns with its well-documented phytochemical composition, notably rich in steroidal saponins, flavonoids, and unsaturated fatty acids such as linoleic and palmitic acids. These compounds are known to compromise bacterial membrane integrity, disrupt metabolic pathways, and induce oxidative stress (Srinivasan, 2006; Thomas et al., 2011; Al-Habori & Raman, 2002; Pandey & Awasthi, 2015). The observed near-complete reduction in E. coli CFUs (99%) and significant inhibition of S. aureus confirms fenugreek’s broad-spectrum efficacy.
Cinnamon’s notable antibacterial activity is consistent with the high concentration of trans-cinnamaldehyde, its principal volatile constituent. Cinnamaldehyde has been shown to inhibit bacterial quorum sensing and biofilm formation, mechanisms essential for pathogenicity and antibiotic resistance (Gill & Holley, 2004; Matan et al., 2006; Chang et al., 2001). The inhibition zone data and CFU reduction strongly support its efficacy, particularly against E. coli, which is in line with prior findings (Friedman et al., 2004).
Carob exhibited the weakest activity among the three, though it still demonstrated measurable antibacterial effects. This result is consistent with its phytochemical profile, which includes high sugar content and comparatively lower levels of polyphenolics and flavonoids. However, the presence of tannins and 5-hydroxymethylfurfural (HMF) may contribute to its moderate inhibitory effects through membrane disruption and oxidative damage (Simsek, 2017; Daglia, 2012; Benković et al., 2018). This suggests possible synergistic action despite the less aggressive antibacterial profile.
Interestingly, all three extracts showed greater activity against E. coli than S. aureus. This could reflect inherent structural differences between Gram-negative and Gram-positive bacteria. While S. aureus possesses a thick peptidoglycan layer, E. coli has an outer membrane rich in lipopolysaccharides that may be more susceptible to phytochemical disruption. Furthermore, efflux pump mechanisms in S. aureus may reduce intracellular accumulation of active compounds (Cowan, 1999).
The observed non-linear dose–response curve, particularly the reduced inhibition zone diameter for fenugreek at 100% concentration, could suggest solubility constraints, compound precipitation, or antagonistic interactions between certain metabolites at higher concentrations (Wagner & Ulrich-Merzenich, 2009). This highlights the importance of considering formulation dynamics and compound synergy when developing plant-based antimicrobial therapies.
Overall, the study reinforces the antimicrobial promise of traditional Sudanese botanicals and supports their potential application as natural antibacterial agents in food safety and therapeutic formulations.
6 Conclusions
All three Sudanese natural products possess antibacterial properties, with fenugreek showing the greatest bactericidal effect and cinnamon a strong bacteriostatic action. Carob, though less potent, still offers measurable inhibition and could serve as a functional food preservative. Future work should (i) fractionate extracts to pinpoint active compounds, (ii) evaluate synergy with standard antibiotics, and (iii) assess cytotoxicity to ensure safety for therapeutic use.
Acknowledgements
The author thanks the Food Analysis and Microbiology Laboratories, University of Gezira, for technical support, and Dr M. Saeed for statistical guidance.
References
Barry, A. L., Garcia, F., & Thrupp, L. D. (1970). Am. J. Clin. Path. 53, 149‑158. Fabricant, D. S., & Farnsworth, N. R. (2001). Environ. Health Perspect. 109(Suppl 1), 69‑75. Srinivasan, K. (2006). Food Rev. Int. 22, 203‑224. Thomas, J. E., et al. (2011). New Biotechnol. 28, 110‑117. Tomaino, A., et al. (2005). Food Chem. 89, 549‑554. Simsek, S. (2017). Food Chem. 221, 191‑198.
Cowan, M. M. (1999). Plant products as antimicrobial agents. Clinical Microbiology Reviews, 12(4), 564–582.
Cushnie, T. P., & Lamb, A. J. (2005). Antimicrobial activity of flavonoids. International Journal of Antimicrobial Agents, 26(5), 343–356.
O’Neill, J. (2016). Tackling drug-resistant infections globally: final report and recommendations. Review on Antimicrobial Resistance.
Prestinaci, F., Pezzotti, P., & Pantosti, A. (2015). Antimicrobial resistance: a global multifaceted phenomenon. Pathogens and Global Health, 109(7), 309–318.
Wagner, H., & Ulrich-Merzenich, G. (2009). Synergy research: approaching a new generation of phytopharmaceuticals. Phytomedicine, 16(2-3), 97–110.
Al-Habori, M., & Raman, A. (2002). Pharmacological properties of fenugreek (Trigonella foenum-graecum L.). Phytotherapy Research, 16(7), 729–736.
Benković, E. T., Knežević, M., Blažeković, B., & Kremer, D. (2018). Antibacterial activity of polyphenolic extracts from carob (Ceratonia siliqua L.) pods. Journal of Herbal Pharmacotherapy, 18(2), 103–112.
Chang, S. T., Chen, P. F., & Chang, S. C. (2001). Antibacterial activity of leaf essential oils and their constituents from Cinnamomum osmophloeum. Journal of Ethnopharmacology, 77(1), 123–127.
Daglia, M. (2012). Polyphenols as antimicrobial agents. Current Opinion in Biotechnology, 23(2), 174–181.
Friedman, M., Henika, P. R., & Mandrell, R. E. (2004). Antibacterial activities of plant essential oils and their components against Escherichia coli O157:H7 and Salmonella enterica in apple juice. Journal of Agricultural and Food Chemistry, 52(19), 6042–6048.
Randhir, R., Lin, Y. T., & Shetty, K. (2004). Phenolics and antioxidant activity in fenugreek (Trigonella foenum-graecum) sprouts. Journal of Food Biochemistry, 28(5), 443–456.

