In Vitro Cytotoxicity Potential of Curcumin Nanoparticles Against Human Cancer Cell Line
الإمكانات السمية الخلوية لجسيمات الكركمين النانوية ضد خط خلايا السرطان البشري
Ibtihal Riyadh Najeeb1
1 Department of pathological analyses,Collage of science,University of Kufa,Iraq.
*Corresponding Author E-mail: ibtihalr.alrammahi@uokufa.edu.iq
DOI: https://doi.org/10.53796/hnsj75/70
Arabic Scientific Research Identifier: https://arsri.org/10000/75/70
Volume (7) Issue (5). Pages: 1249 - 1257
Received at: 2026-04-15 | Accepted at: 2026-04-22 | Published at: 2026-05-01
Abstract: Breast cancer remains a major global health challenge, and the search for safer and more effective anticancer agents has encouraged the development of nanoparticle-based drug delivery systems. Curcumin is a natural polyphenolic compound with recognized anticancer properties; however, its clinical application is limited by poor solubility and low bioavailability. Therefore, this study aimed to synthesize and evaluate curcumin nanoparticles as a potential anticancer formulation against the human breast cancer cell line MCF-7. Curcumin nanoparticles were prepared using a modified sol–oil method and characterized through morphological and physicochemical techniques, including SEM, AFM, XRD, FTIR, and UV–visible spectroscopy. The synthesized nanoparticles appeared as a stable yellow powder with improved dispersibility compared with raw curcumin. Morphological analysis confirmed the formation of spherical to quasi-spherical nanoparticles with nanoscale dimensions and relatively uniform distribution. Spectroscopic and structural analyses further indicated that the characteristic chemical structure of curcumin was preserved after nanoparticle formation. The anticancer activity of the prepared nanocurcumin was assessed using the MTT assay against MCF-7 cells after 72 hours of exposure at different concentrations. The results demonstrated a clear concentration-dependent cytotoxic effect, with inhibition rates of 35.47%, 49%, 57.95%, 71.9%, and 79.66% at concentrations of 6.25, 12.5, 25, 50, and 100 µg/mL, respectively. The estimated IC₂₀ value was approximately 14 µg/mL, indicating notable antiproliferative activity. These findings suggest that nanoscale formulation enhances the biological effectiveness of curcumin, likely through improved dispersibility, solubility, and cellular uptake. Overall, the prepared curcumin nanoparticles showed promising in vitro cytotoxic potential against breast cancer cells and may represent a useful candidate for further anticancer drug development. Additional studies are recommended to clarify the molecular mechanisms involved and to evaluate the safety and therapeutic efficacy of this formulation in vivo.
Keywords: Curcumin nanoparticles; Nanocurcumin; MCF-7 cell line; Breast cancer; Cytotoxicity assay.
المستخلص: يُعد سرطان الثدي من أبرز التحديات الصحية العالمية، وقد شجّع البحث عن عوامل مضادة للسرطان أكثر أمانًا وفاعلية على تطوير أنظمة توصيل دوائية قائمة على الجسيمات النانوية. ويُعد الكركمين مركبًا بوليفينوليًا طبيعيًا يمتلك خصائص معروفة مضادة للسرطان؛ إلا أن تطبيقه السريري يظل محدودًا بسبب ضعف ذوبانيته وانخفاض توافره الحيوي. لذلك، هدفت هذه الدراسة إلى تحضير جسيمات الكركمين النانوية وتقييمها بوصفها صيغة محتملة مضادة للسرطان ضد خط خلايا سرطان الثدي البشري MCF-7.حُضّرت جسيمات الكركمين النانوية باستخدام طريقة السول–زيت المعدّلة، وتم توصيفها من خلال تقنيات مورفولوجية وفيزيوكيميائية شملت المجهر الإلكتروني الماسح، والمجهر القوة الذرية، وحيود الأشعة السينية، ومطيافية الأشعة تحت الحمراء، ومطيافية الأشعة فوق البنفسجية–المرئية. ظهرت الجسيمات النانوية المحضّرة على هيئة مسحوق أصفر مستقر يتميز بقابلية تشتت أفضل مقارنة بالكركمين الخام. وأكد التحليل المورفولوجي تكوّن جسيمات نانوية كروية إلى شبه كروية ذات أبعاد نانوية وتوزيع متجانس نسبيًا. كما أشارت التحليلات الطيفية والبنيوية إلى احتفاظ الكركمين بتركيبه الكيميائي المميز بعد تحويله إلى الصيغة النانوية. وتم تقييم النشاط المضاد للسرطان للنانوكركمين المحضّر باستخدام اختبار MTT ضد خلايا MCF-7 بعد 72 ساعة من التعرض لتراكيز مختلفة. أظهرت النتائج تأثيرًا سميًا خلويًا واضحًا معتمدًا على التركيز، إذ بلغت نسب التثبيط 35.47% و49% و57.95% و71.9% و79.66% عند التراكيز 6.25 و12.5 و25 و50 و100 ميكروغرام/مل على التوالي. كما بلغت قيمة IC₂₀ المقدّرة نحو 14 ميكروغرام/مل، مما يشير إلى نشاط ملحوظ مضاد للتكاثر. وتشير هذه النتائج إلى أن الصياغة النانوية تعزز الفاعلية الحيوية للكركمين، ويرجح أن ذلك يعود إلى تحسن قابليته للتشتت والذوبانية والامتصاص الخلوي. وبوجه عام، أظهرت جسيمات الكركمين النانوية المحضّرة إمكانات واعدة في السمية الخلوية المختبرية ضد خلايا سرطان الثدي، وقد تمثل مرشحًا مفيدًا لمزيد من التطوير كعامل مضاد للسرطان. وتوصي الدراسة بإجراء بحوث إضافية لتوضيح الآليات الجزيئية المعنية وتقييم سلامة هذه الصيغة وفاعليتها العلاجية داخل الجسم الحي.
الكلمات المفتاحية: جسيمات الكركمين النانوية؛ النانوكركمين؛ خط خلايا MCF-7؛ سرطان الثدي؛ اختبار السمية الخلوية.
1. Introduction
If you are training on a model that does not train on data past October 2023, cancer is still one of the top causes of death in the world. World Health Organization (WHO) estimates state that 8.2 million deaths were caused by cancer in the year 2012, and more than 13 million deaths, occurring until the year of 2030 (Chang et al., 2017 and Ahmed & Guda, 2025). It is likely the result of drug resistance, tumor recurrence, severe side effects and the general therapeutic nonspecificity associated with traditional cancer treatments (i.e. chemotherapy and radiation therapy). Therefore, the emergence of a multitude of breakthrough innovations for safer and more effective treatment infrastructures has been ranked as one of the most critical focal points in cancer research (Liu et al., 2025). Due to the distinct physical and chemical characteristics which nanomaterials possess, recent developments in cancer therapies have investigated the use of nanoparticles as a promising tool offered by nanotechnology during treatment of cancers. Nanoparticles (1 nm–100 nm) increase the solubility, stability, and targeted distribution to tumor tissues, which are a unique advantage of nanocarriers over drug delivery (Alhasnawi et al., 2025). Nanotechnology can be employed in various fields; however, biomedical and pharmaceutical sciences are two very important fields. Of those, the sol-gel method of generating nanoparticles has gained much interest among various nanosystems since its ease of production process only requires using non-toxic chemicals that may affect future biomedical uses (Alsultan & Guda, 2026). Curcumin is a naturally occurring polyphenolic compound extracted from the roots of the turmeric plant (Curcuma longa). It has attracted much attention in the literature because of its various biological effects, including antioxidant, anti-inflammatory, antibacterial, and anticancer characteristics (Amaroli et al., 2024 and Hanan et al., 2025). Nanocurcumin can accomplish more pronounced therapeutic properties than natural curcumin owing its high solubility, bioavailability, cellular uptake efficiency with lower doses and greater targeting to affected tissues (Wen et al., 2017 and Saba et al., 2025). The treatment of small doses of nanocurcumin is more effective on many human cancer cell lines (Basniwal et al., 2014). The present study will synthesize this nanocurcumin using oil-in-water emulsion method to enhance its solubility, absorption, and bioavailability. Then cytotoxicity and anticancer activity of the compound against human breast cancer cells (MCF-7) will be determined using MTT assay, as a potential conventional therapy alternative.
2. Materials and methods
2.1. Synthesis of curcumin nanoparticles
They are prepared using a modified oil-sol method based on the original study (hanna and Saad, 2020).Curcumin (10 mg) was dissolved in 0.1 mL DMSO and added to 10 mL olive oil under stirring at 600 rpm for 1 h. Next, the solution was sonicated for 2 h at 80 C under ultrasonic conditions, where the sonication amplitude was 5 mm and the pulses were 4 s long with an interval of 1 min between successive pulses. The resulting mixture was directly frozen in liquid nitrogen for 10 min and was then incubated at room temperature for 4 h. The particles formed were obtained by centrifugation at 6000 rpm for 10 min at 4 C, followed by washing with diethyl ether and then dissolved in distilled water and finally freeze-dried to obtain nanocurcumin powder.in nanoparticles (nanocurcumin).
2.2. Characterization of the prepared nanocurcumin
Atomic force microscopy and field emission scanning electron microscopy (FESEM) were used to study the morphological properties of the produced curcumin nanoparticles, as well as X-ray diffraction, infrared spectra, ultraviolet and visible spectra.
2.3. Anticancer activity of curcumin nanoparticles
2.3.1. Maintenance of cell cultures
At the AL-Amin Center for Advanced Biotechnology Research and Techniques in Najaf, the MCF-7 cell line was kept in RPMI-1640 supplemented with 10% fetal bovine, 100 units/mL penicillin, and 100 µg/mL streptomycin. Trypsin-EDTA was used to passage the cells, which were then reseeded twice a week at 80% confluence and cultured at 37 °C (Sulaiman et al., 2018).
2.3.2. Cytotoxicity assays of curcumin nanoparticles
Using 96-well plates, the MTT cell viability assay was used to assess the cytotoxic effect of curcumin nanoparticles. The cells were exposed to the prepared curcumin nanoparticles at different concentrations. After 24 hours and after 72 hours of treatment, the media were removed, 28 microliters of MTT at a concentration of 2 mg/ml were added, and the cells were successfully maintained in the incubator at 37°C. Following the removal of the MTT solution, 130 µL of DMSO (dimethyl sulphoxide) was added to the wells to dissolve the residual crystals. The mixture was then incubated at 37 °C for 15 minutes while being shaken (Jabir et al., 2019). The assay was carried out in triplicate, and the absorbance was measured at a wavelength of 492 nm using an ELISA plate reader. The following formula was used to determine the percentage of cytotoxicity, or the inhibition rate of cell growth:
Cytotoxicity = A-B/A *100……………………….1
Where A the optical density of control and B the optical density of Samples.
3. Results and discussion
3.1. Synthesis of Curcumin nanoparticles
The modified sol-oil approach was successfully used to create curcumin nanoparticles. The combination gradually changed from a clear yellow solution to a turbid yellow suspension during the preparation procedure, signifying the creation of curcumin nanoparticles. A fine yellow precipitate was produced following centrifugation and washing, indicating that nanocurcumin was successfully formed. When compared to raw curcumin, the drying process produced a stable yellow powder that could be redispersed, indicating successful nanoparticle production and enhanced dispersibility.The fast diffusion of curcumin from the organic phase into the oil phase under constant stirring is the primary cause of nanoparticle production in the sol–oil technique. While sonication improves dispersion, decreases particle size, and minimizes aggregation to produce stable nanoscale particles, the use of a hot plate stirrer guarantees uniform mixing.These findings are consistent with earlier research demonstrating that curcumin nanoparticles made using oil-based techniques had superior biological activity and solubility compared to free curcumin. The produced nanocurcumin demonstrated enhanced dispersion and nanoscale particle size, which may improve its cellular uptake and potential for cancer treatment (Hanna and Saad, 2020).
3.2. Morphological characterization of the synthesized Curcumin nanoparticles
Figure 1 shows the atomic force microscopy examination of curcumin nanoparticles. The image above shows the bright white and yellow dots, which represent curcumin nanoparticles distributed on the surface. The XY dimensions indicate that the scan is 10 by 10 meters. The colored bar on the right shows the vertical height of the particles. The highest point recorded in this scan is 3.5 nanometers. This indicates that the particles are very thin or almost flat on the surface, which is a good indicator of size. The lower graph shows the histamine size, where the curved approach shows a very sharp beige peak close to zero, usually between 1 and 4 nanometers, depending on the scale. The narrowness of the curve indicates that the particles are very homogeneous in size, meaning that most of the prepared curcumin particles are approximately the same size and not a mixture of large and small particles. Figure 2 (FESEM) shows the curcumin nanoparticles, revealing a quasi-spherical structure with a uniform distribution. The surface morphology exhibits a coarse texture, likely due to the presence of bioactive organic compounds from the plant extract on the nanoparticle surface. The particle size is 100 nm. The distinct crystalline lattice edges indicate their crystalline nature, as shown in Figure 3. X-ray diffraction (XRD) patterns showed characteristic diffraction peaks at 2θ values of 25–27. The average crystal size, calculated using the Debye–Scherer equation, ranged between 23 and 25 nm, confirming the nanoparticle nature. Figure 4 shows of FTIR, The broad peak (~3400–3500 cm⁻¹): represents the phenolic (O–H) group. In nanoparticles, this peak is often sharpened or slightly altered due to the increased surface area of the molecules, which increases the exposure of hydrogen bonds. The peak at (~1625 cm⁻¹): the most prominent in your image, is due to the overlapping (C=O) and (C=C) double bond vibrations. The fact that this peak remains sharp indicates that the dictone structure of curcumin is unaffected.The peak at (~1510 cm⁻¹): represents the aromatic (C=C) benzene ring vibrations.The peak at (~1270 cm⁻¹): represents the stretching of the (C–O) phenolic bond, a very distinctive marker of curcumin purity.The peak at (~1020–1150 cm⁻¹): represents the methoxy (C–O–C) bond vibrations. Figure 5 shows the ultraviolet and visible spectrum. A clear absorption peak is observed at a wavelength of 420 nm. Nano-curcumin is known for its strong absorption in the blue region of the visible spectrum, between 400-430 nanometers. This explains its orange-yellow color to the human eye. This value confirms that the substance is indeed curcumin and that its chemical structure, specifically the system of alternating double bonds, remains intact after the transformation to the nanoscale. Nano-curcumin often exhibits a slight shift in the apex, with an increase in apex width, compared to regular curcumin.
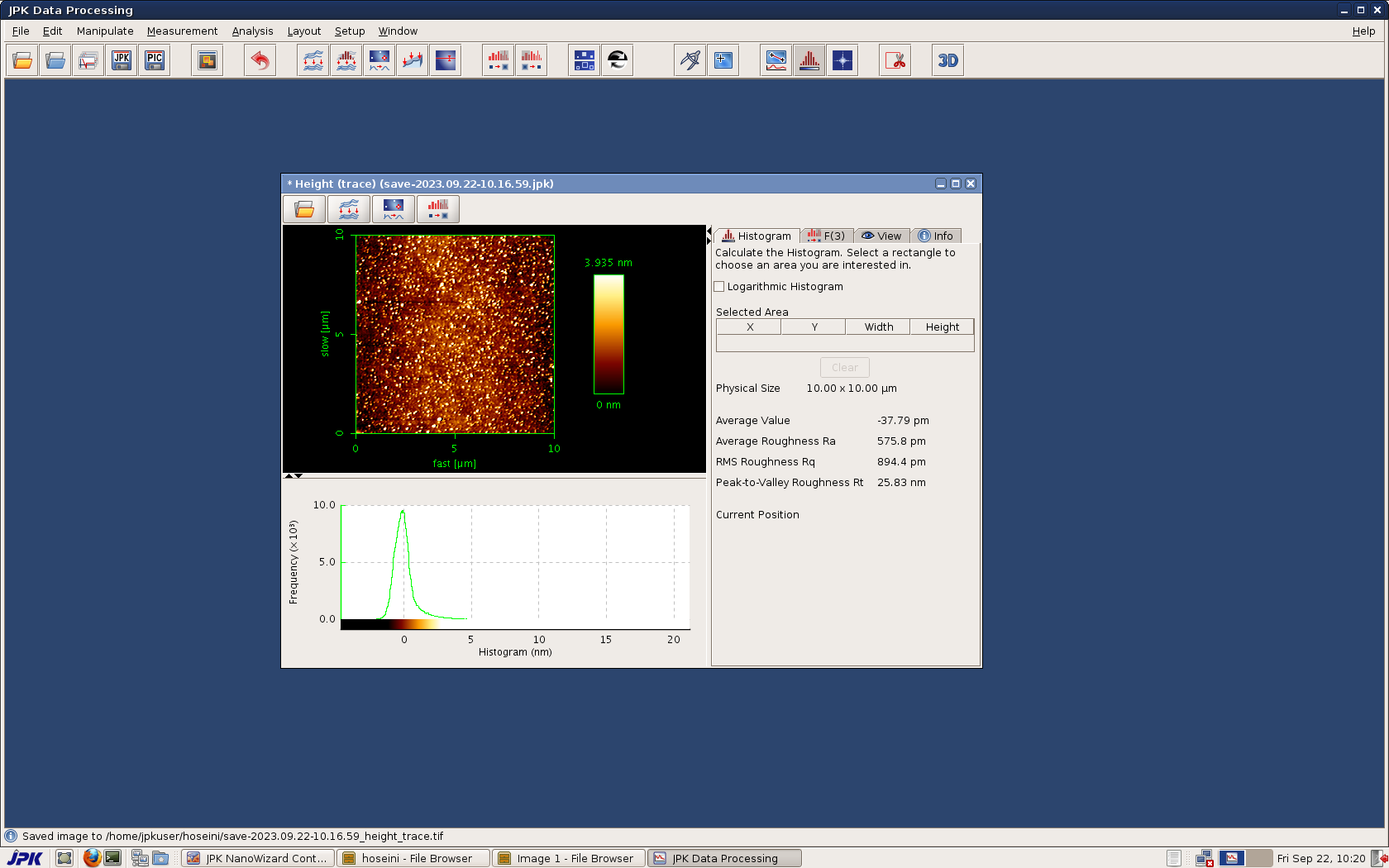
Figure 1: Atomic force microscopy of Curcumin nanoparticles

Figure 2 : FESEM of Curcumin nanoparticles
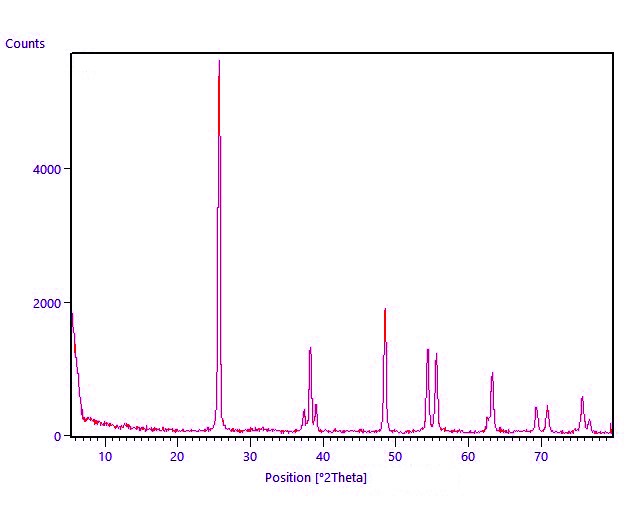
Figure 3: (XRD) spectra of Curcumin nanoparticles
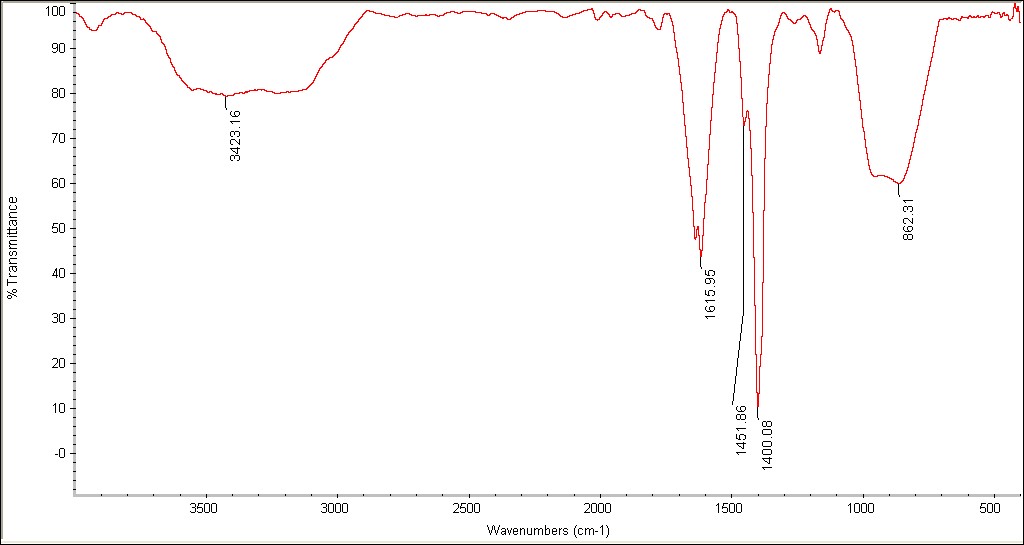
Figure 4: FTIR of Curcumin nanoparticles
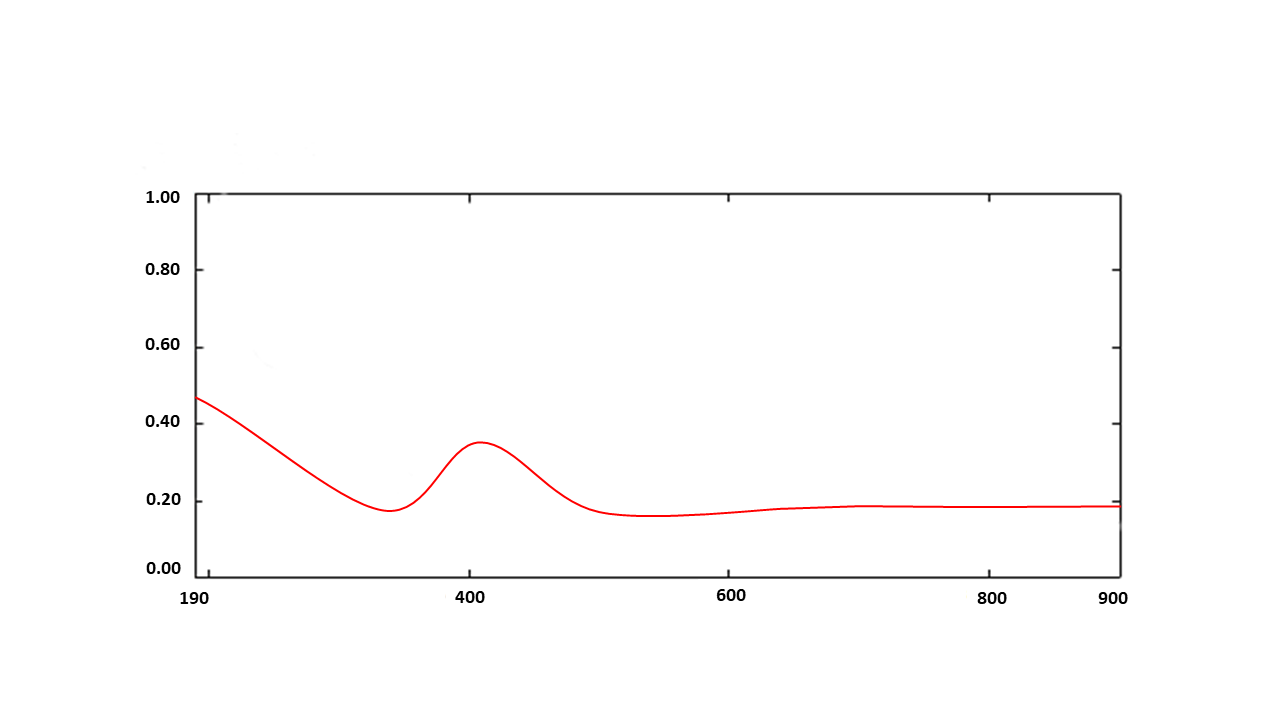
Absorbance a.u.
Wavelength.nm
Figure 5: UV of Curcumin nanoparticles
3.3. Anticancer activity (MTT assay)
3.3.1. Cytotoxicity effect of Curcumin nanoparticles on human of MCF-7 cell
The MTT assay is a colorimetric test that uses cell metabolic activation to cause a color shift. For 72 hours, the cytotoxic effects of curcumin nanoparticles on human cancer cell lines (MCF-7) were investigated. After 72 hours, MCF-7 proliferation was significantly inhibited, according to the data. When compared to the untreated control cells, the cell growth was noticeably reduced.For 72 hours, the cells were exposed to varying doses of curcumin nanoparticles (6.25, 12.5, 25, 50, and 100 µg/mL). After 72 hours of exposure, curcumin nanoparticles exhibited 35.47%, 49%, 57.95%, 71.9%, and 79.66% of the cytotoxicity at 6.25µg/ml, 12.5µg/ml, 25µg/ml, 50µg/ml, and 100µg/ml, respectively. After 72 hours of treatment, curcumin nanoparticles’ estimated IC•₀ value against MCF-7 cells was roughly 14 µg/mL. (Fig. 6a,b). These results support earlier findings that nanocurcumin has more robust antiproliferative effects than free curcumin in breast cancer models (Elbialy et al., 2013) by confirming that increasing nanoparticle concentrations results in higher inhibition of breast cancer cell growth.The anticancer efficacy of nanocurcumin against MCF-7 cells is strongly supported by studies focused on breast cancer. According to Hosseini et al. (2019), nano-curcumin decreased MCF-7 cell viability by roughly 83.6% at their maximum measured concentration (162.87 mmol/L = 60 mg/mL), with an IC•¹ value of 59.72 mmol/L ≈ 22 mg/mL. Nanocurcumin showed a similar cytotoxic impact (79% vs. 83.6%) despite the much lower dose utilized in this investigation (100 µg/mL) compared to Hosseini et al. (≈ 60 mg/mL), demonstrating robust antiproliferative action even at significantly lower concentrations. According to Tsai et al. (2025), showed that after 72 hours, polysaccharide-based curcumin nanoparticles showed high cytotoxic effects against MCF-7 cells (IC²₀ = 17.5 µg/mL), with quick cellular uptake and apoptosis induction.These results are in line with earlier research showing that nanocurcumin has comparable cytotoxic and antiproliferative effects on breast cancer cells. Moreover, similar dose- and time-dependent cytotoxic effects have been seen in different cancer models. According to Hanna and Saad’s (2020) report, the cytotoxicity of nanocurcumin on Hep-2 cells increases with concentration and exposure time, reaching approximately 97.8% cell death at 75µg/mL after 48 hours. The IC₀ values decreased from 45.1 µg/ml after 24 hours to 17 µg/ml after 48 hours. This highlights the interplay between exposure time and dose in determining cytotoxic activity. In a similar study, Alam et al. (2022) demonstrated that curcumin-PLGA nanoparticles inhibited proliferation of human gastric cancer cells (AGS line) by 97% at a concentration of 40 µM over 72 hours, whereas free curcumin exhibited only 83% inhibition at the same concentration. The curcumin nanoparticles (mean IC21 hours) were found to have an IC₂¹ 18.78 µM, whereas free curcumin (6-hours) had a 24.20 µM; thus the two formulations show that the nanoparticle formulation helps increase cytotoxic activity. Extended exposure only permits nanoparticle interaction with biological structures, which in turn disrupts cellular metabolism and prevents cell development. Conclusions Our data support the utility of nanocurcumin as a potent anticancer drug with therapeutic flexibility for clinical functioning. Moreover, analogous results have been observed in other studies showing selectivity towards normal cells and some cancer cell types that appear to function in a manner parallel to the one demonstrated here (Karimi et al., 2024). (2020); Moawad (et al.) (2023)and Alshebly & Guda (2026). Numerous in vitro investigations have demonstrated that nanocurcumin is more effective than free curcumin at increasing cytotoxicity and apoptosis in cancer cells, as demonstrated by increased pro-apoptotic gene expression, disruption of cell cycle progression, and accelerated programmed cell death.
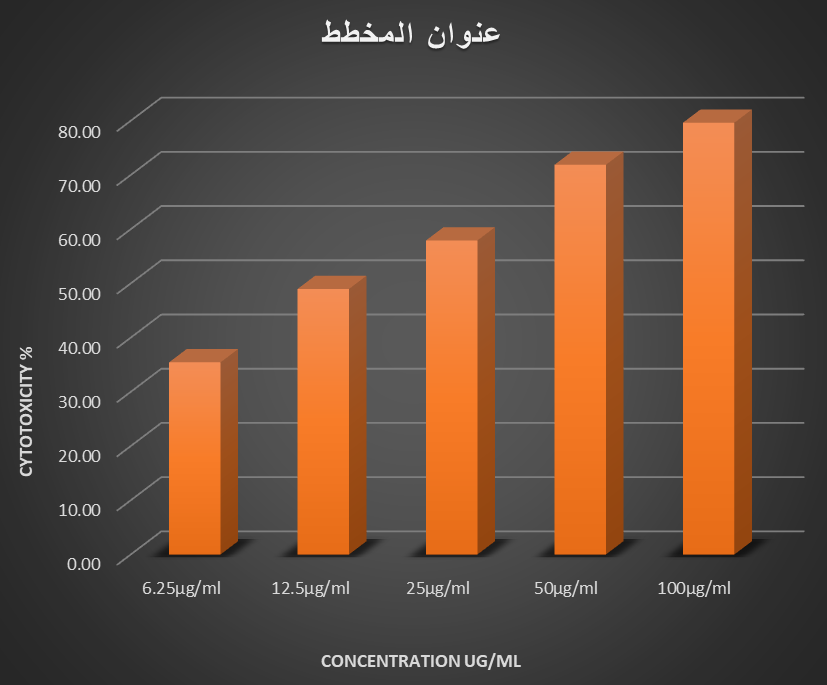 .
.
a
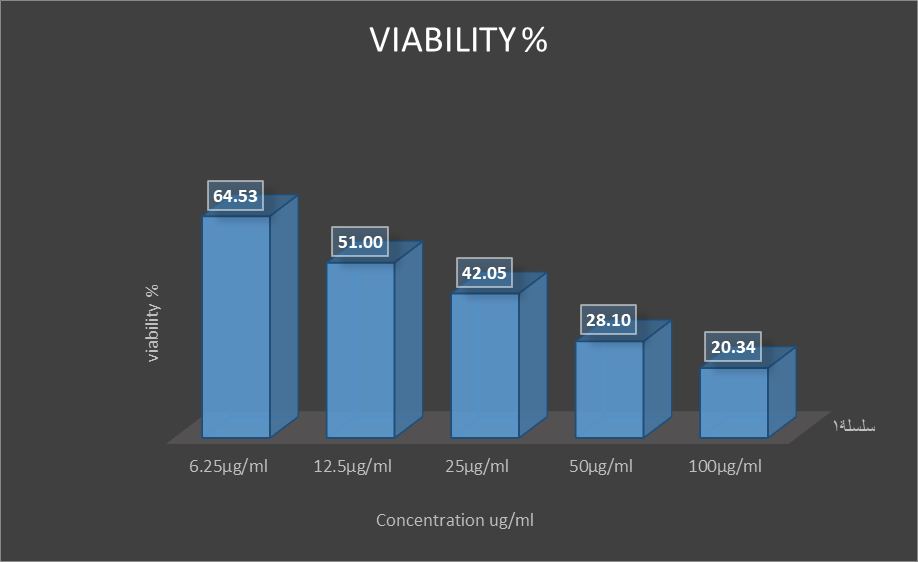
b
Figure 6: Anticancer activity (MTT assay) of Curcumin nanoparticles
4. Conclusions
The sol–oil process was effectively used to create curcumin nanoparticles, producing a stable and evenly distributed nanosystem. Despite minor aggregation, SEM investigation verified spherical to quasi-spherical morphology with nanoscale size and comparatively uniform dispersion. The MTT test results showed that nanocurcumin significantly inhibits the growth of MCF-7 breast cancer cells in a concentration-dependent manner.These results demonstrate the potential of nanocurcumin as a potent anticancer drug.
References
Ahmed, A. Q., & Guda, M. A. (2025). Comparison and characterization of green synthesized silver oxide nanoparticles using Ziziphus spina-christi plant extract and AgO NPs by femtosecond laser ablation technique. Contemporary Research Analysis Journal, 2(12), 762–769. https://doi.org/10.55677/CRAJ/02-2025-Vol02I12
Alam, J., Dilnawaz, F., Sahoo, S. K., Singh, D. V., Mukhopadhyay, A. K., Hussain, T., & Pati, S. (2022). Curcumin encapsulated into biocompatible co-polymer PLGA nanoparticle enhanced anti-gastric cancer and anti-Helicobacter pylori effect. Asian Pacific journal of cancer prevention: APJCP, 23(1), 61.
Alhasnawi Radhi Jubair Nasser, Guda Muslim Abd Muthik, Al-Bayati Sami Ammar (2025). Effect of Nano-silver Fertilization on the Tolerance of Salt Stress by Irrigation with Saline Water in Faba Bean (Vicia faba L.) . Indian Journal of Agricultural Research. (): . doi: 10.18805/IJARe.AF-968.
Alshebly, E. A. J., & Guda, M. A. (2026, March). Green synthesis of silver oxide nanoparticles and their effect on inhibition growth of bacteria. In AIP Conference Proceedings (Vol. 3396, No. 1, p. 030008). AIP Publishing LLC. https://doi.org/10.1063/5.0318556
Alsultan, H. K. A. A. K., & Guda, M. A. (2026, March). Green synthesis and characterization of zirconium oxide nanoparticles using Matricaria Chamomilla L. plant extracts for antibacterial application. In AIP Conference Proceedings (Vol. 3396, No. 1, p. 080001). AIP Publishing LLC. https://doi.org/10.1063/5.0318554
Amaroli, A. (2024). The bright side of curcumin: A narrative review. Cancers, 16(14), 2580.
Basniwal, R. K., Khosla, R., & Jain, N. (2014). Nutrition and Cancer, 66(6), 1015–1022.
DOI: 10.55677/CRAJ/05-2025-Vol02I08
Elbialy, N. et al. (2013). Cytotoxicity of nanocurcumin on cancer cells. Journal of Applied Research
Eqbal Yousif Abed1, Hanan Fadhil Kadhim2, Muthik A. Guda .(2025)The Effects of Climate Change on Public Health, Contemporary Research Analysis Journal,8(2),p 500-503.
G.M. Sulaiman, M.S. Jabir, A.H. Hameed. Nanoscale modification of chrysin for improved of therapeutic efficiency and cytotoxicity. Arificial cells, Nanomedicine, and biotechnology, (2018),1-8.
Gandapu, U., Chaitanya, R., Kishore, G., & Kondapi, A. (2011). Curcumin-loaded nanoparticles: preparation and characterization. PLoS ONE.
Ghoran, S. H., Calcaterra, A., Abbasi, M., et al. (2022). Curcumin-based nanoformulations: A promising adjuvant towards cancer treatment. Molecules, 27(16), 5236.
Hamad, M. O., Rahi, A. Y., & Guda, M. A. (2023). Study of Staphylococcus spp. isolated from Clinical Cases and determination of its multidrugresistant to antibiotics and phytosynthesized ZnO (NPs). Cardiometry, (26), 227-235.
Hammad, M. O., & Guda, M. A. (2023). Effect of Phytosynthesis silver oxide nanoparticles on multidrug-resistant Klebsiella pneumonia isolated from children. Eur. Chem. Bull, 12(1), 1655-1669.
Hanan Fadhil Kadhim1, Eqbal Yousif Abed2, Muthik A. Guda3.(2025). Climate Change and Its Influence on Anatomical and Physiological Features of Pomegranate (Punica Granatum L.) Leaves, Contemporary Research Analysis Journal 8(2)p: 510-515
Hanna, D. H., & Saad, G. R. (2020). Retracted Article: Nanocurcumin: preparation, characterization and cytotoxic effects towards human laryngeal cancer cells. RSC advances, 10(35), 20724-20737.
Hosseini, S., Chamani, J., Hadipanah, M. R., Ebadpour, N., Hojjati, A. S., Mohammadzadeh, M. H., & Rahimi, H. R. (2019). Nano‑curcumin’s suppression of breast cancer cells (MCF‑7) through the inhibition of cyclinD1 expression. Breast Cancer: Targets and Therapy, 11, 137–142. https://doi.org/ 10.18805/IJARe.AF-968
Karimi, M., Qomi, M., Hadipour Jahromy, M., Parsania, M., & Motakef Kazemi, N. (2024). Preparation and evaluation of Curcumin nano emulsion to inhibit TC-1 cell growth. Iranian Journal of Chemistry and Chemical Engineering, 43(11), 3879-3892.
Liew, K. B., Phang, H. C., Tan, V. Y. X., et al. (2025). Nanoparticles as novel drug delivery systems for cancer treatment: Current status and future perspectives. Current Drug Delivery, 31(39), 3117–3127.
M.S. Jabir, A.A. Taha, U.I. Sahib, Z.J. Taqi, A.M. Al-Shammari, A.S. Salman, Novel of nano delivery system for linalool loaded on gold nanoparticles conjugated with CALNN peptide for application in drug uptake and induction of cell death on breast cancer cell line, Materials science and engineering C, 94(2019) 949-964.
Mahmoud Moawad, Nasr, G. M., Osman, A. S., & Shaker, E. S. (2023). Curcumin nanocapsules effect in apoptotic processes, gene expression, and cell cycle on Hep‑G2 cell lines. International Journal of Immunopathology and Pharmacology, 37.
Nasser Jubair Radhi Alhasnawi1, Muthik Abd Muslim Guda, Ammar Sami Al-Bayat. (2025). Effect of Nano-silver Fertilization on the Tolerance of Salt Stress by Irrigation with Saline Water in Faba Bean (Vicia faba L.). Indian Journal of Agricultural Research.
Saba Abd Al-Mutleb Hamood1, Douaa Abdulrazzaq Khaleel2, Muthik A Guda,(2025), Harnessing Artificial Intelligence to Revolutionize Public Health: Innovations in Prevention, Monitoring, and Policy Decision-Making, Contemporary Research Analysis Journal,8(2),p: 504-509. DOI: 10.55677/CRAJ/06-2025-Vol02I08
Tsai, Y.‑C., Miyajima, H., Chou, M.‑Y., & Fujita, S. (2025). Curcumin‑Loaded Polysaccharide Nanoparticles Enhance Aqueous Dispersibility and In Vitro Cytotoxicity in Breast Cancer Cell Lines. Nanomaterials, 15(22), 1747.
Wen, C., Zhou, Y., Zhou, C., Zhang, Y., Hu, X., Li, J., & Yin, H. (2017). Journal of Nanomaterials, 2017, 8.
Zhang, Y.; Liang, Y. and He, C. (2017). Anticancer activities and mechanisms of heat-clearing and detoxicating traditional Chinese herbal medicine. .Chin. med., 12(1), 20.

