Numerical Study of Flame Structure and Precursors Formation of propane–H2 Mixtures
دراسة عددية لبنية اللهب وتكوّن المركبات الأوّلية في خليط البروبان–الهيدروجين
Nasreldin M. Mahmoud1,*
1 Mechanical Engineering Department, Faculty of Engineering, University of Sinnar, Sinnar, Sudan
*Corresponding author. Mechanical Engineering Department, Faculty of Engineering, University of Sinnar, Sinnar, Sudan
E-mail address: nasrmohamed53@hotmail.com
DOI: https://doi.org/10.53796/hnsj612/37
Arabic Scientific Research Identifier: https://arsri.org/10000/612/37
Volume (6) Issue (12). Pages: 549 - 557
Received at: 2025-11-10 | Accepted at: 2025-11-18 | Published at: 2025-12-01
Abstract: Combustion of propane-hydrogen mixtures represents a promising strategy for reducing soot and polycyclic aromatic hydrocarbon (PAH) emissions while taking advantage of existing fuel infrastructure. This study numerically investigates the effects of H2 addition (10–30 % by mole) on the flame structure and formation of key aromatic precursors in a co‑flow propane diffusion flame using ANSYS Fluent with a detailed chemical mechanism. Fictitious inert specie (XH₂) was used to isolate chemical effects. The numerical Results indicated that the addition of H2 reduces the peak flame temperature by up to 19 K, primarily due to its higher specific heat capacity and the upstream shift of the reaction zone. While the chemical effect of H2 promotes flame temperature due to increased free radical concentrations. In addition, the addition of H2 accelerates oxidation kinetics and inhibits the formation of benzene (A1) and its precursors, acetylene (C₂H₂) and propargyl (C₃H₃). The chemical inhibition of A1 becomes more pronounced with increasing H2 addition ratios. These results provide key insights into the chemical role of H2 in the propane flame, and support the improvement of propane-hydrogen mixtures for cleaner combustion systems.
Keywords: Hydrogen; Propane; Coflow diffusion flame; Aromatic formation.
المستخلص: يمثّل احتراق خلطات البروبان–الهيدروجين استراتيجية واعدة للحد من انبعاثات السخام والهيدروكربونات العطرية متعددة الحلقات (PAHs)، مع الاستفادة في الوقت نفسه من البنية التحتية الحالية للوقود. تهدف هذه الدراسة إلى التحقيق العددي في تأثير إضافة الهيدروجين (بنسب تتراوح بين 10–30% مولياً) على بنية اللهب وتكوّن السلائف العطرية الرئيسة في لهب انتشار للبروبان ذي الجريان المشترك، باستخدام برنامج ANSYS Fluent مع آلية كيميائية تفصيلية. وقد استُخدم نوع خامل افتراضي (XH₂) لعزل التأثيرات الكيميائية. أشارت النتائج العددية إلى أن إضافة الهيدروجين تؤدي إلى خفض درجة حرارة الذروة في اللهب بما يصل إلى 19 كلفن، ويُعزى ذلك أساساً إلى سعته الحرارية النوعية الأعلى وإلى انتقال منطقة التفاعل نحو المنبع. في المقابل، يُسهم التأثير الكيميائي للهيدروجين في تعزيز درجة حرارة اللهب نتيجة زيادة تراكيز الجذور الحرة. إضافة إلى ذلك، تعمل إضافة الهيدروجين على تسريع حركيات الأكسدة وتثبيط تكوّن البنزين (A1) وسلائفه، وهما الأسيتيلين (C₂H₂) والبروبارجيل (C₃H₃). ويزداد تأثير التثبيط الكيميائي لتكوّن البنزين (A1) بازدياد نسبة إضافة الهيدروجين. توفر هذه النتائج فهماً أساسياً للدور الكيميائي للهيدروجين في لهب البروبان، وتدعم تطوير خلطات البروبان–الهيدروجين من أجل أنظمة احتراق أنظف.
الكلمات المفتاحية: الهيدروجين؛ البروبان؛ لهب الانتشار ذو الجريان المشترك؛ تكوُّن المركبات العطرية.
Introduction
The global energy landscape is undergoing a significant transformation driven by the urgent need to reduce harmful emissions and improve air quality [1]. The combustion of conventional hydrocarbon fuels remains a primary source of energy. In the meanwhile, it is a major contributor to carbon dioxide (CO₂) and pollutant emissions, including soot and polycyclic aromatic hydrocarbons (PAHs) [2, 3]. The negative impacts of PAHs emissions on human health and the environments necessitate the development of advanced clean combustion technologies[4-7]. By using clean carbon-free nature fuel and thus reducing aromatic content is an effective approach to inhibit soot and PAHs formation in both internal combustion engines and industrial boilers [8].
hydrogen (H₂) has emerged as a pivotal energy carrier due to its high energy content per mass and its carbon-free nature, resulting in zero CO₂ emissions from combustion [9]. However, challenges related to hydrogen storage, safety, and flame stability have motivated the strategy of blending hydrogen with existing hydrocarbon fuels. This approach leverages hydrogen’s advantages, such as its high laminar flame speed and wide flammability limits, to enhance combustion efficiency and reduce carbon-based emissions, serving as a practical transition towards a sustainable hydrogen economy [10].
Propane (C₃H₈), a primary component of liquefied petroleum gas (LPG), is a widely used fuel in domestic, industrial, and transportation sectors. Its established infrastructure and relatively clean combustion profile make it an excellent candidate for hybridization with hydrogen [11]. The addition of hydrogen to propane can fundamentally alter the flame structure, temperature distribution, and chemical reaction pathways. Notably, the impact of H₂ on soot and PAH precursors is complex and highly dependent on the base fuel [12-18]. For instance, studies on methane and ethylene flames have shown that H₂ addition can sometimes promote soot formation by enhancing the concentrations of key precursors like acetylene (C₂H₂) and benzene (A1) [19, 20]. In contrast, numerical studies on n-dodecane flames have indicated that H₂ can suppress the formation of A1 and pyrene (A4), leading to reduced soot formation [21]. This fuel-specific behavior underscores the necessity of dedicated studies for propane-hydrogen mixtures.
Despite that numerous studies have focused on small C1-C2 hydrocarbon fuel flames, a detailed investigation into the effect of H₂ addition on the formation of aromatic species in propane diffusion flames remains less explored. Furthermore, the fuel inlet boundary state, including composition and velocity, plays a critical role in determining the flame’s structure and the subsequent formation of pollutants [22]. Therefore, this work presents a numerical study of a co-flow diffusion flame to systematically investigate the effects of hydrogen blending on the flame structure and the formation of PAH precursors in propane flames. The result is expected to build accurate kinetic understanding to aid in the optimization of practical combustion systems utilizing propane-hydrogen blends.
Numerical Methodology
The numerical simulations in this work were performed using the ANSYS Fluent software package [23]. A detailed chemical mechanism was employed to accurately capture the pyrolysis and oxidation processes [24]. This machoism comprising 533 chemical reactions and 99 species up to A4. 2D-axisymmetric, pressure-based solver was used under steady-state conditions. The governing equations for mass, momentum, energy, and species transport were solved based on based on coupled algorithm. Non-uniform meshes were adopted for simulating co-flow burners which provides a good balance between accuracy and computational cost for this configuration [25-27]. The computational domain was designed to represent a standard co-flow diffusion flame burner as in previous work [25-27] , with a fuel tube diameter of 1.08 cm and a co-flow air diameter of 6 cm. Non-uniform meshes were adopted for simulating co-flow burners which provides a good balance between accuracy and computational cost for this configuration [25-27]. In detail, non-uniform grid with approximately 6000 cells was generated, with significant mesh refinement in the flame zone to resolve high gradient regions accurately. Grid independence was confirmed by comparing the temperature and major species profiles with a finer grid of 9000 cells, showing negligible differences (<1% in peak temperature). A mixture of propane and hydrogen (with H₂ mole fractions of 10%, 20%, and 30%) was introduced at a temperature of 298 K. The inlet velocity was assumed as a parabolic velocity profile for the fuel stream while a uniform profile of 6.2 cm/s was specified for the air Co-flow Inlet The simulation was considered converged when the residuals for all variables fell below 10⁻⁶.
Results and Discussion
The numerical results provide detailed insight into how H2 addition alters the fundamental characteristics of a propane co-flow diffusion flame, with significant implications for its chemical effects on precursor and aromatic species formation.
3.1 Flame Structure and Temperature Distribution with H2 addition
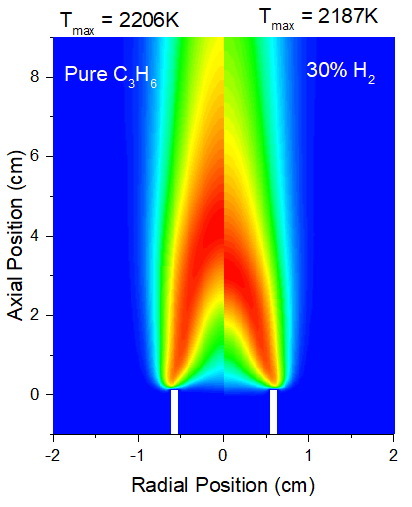
As depicted in Figures 1 and 2, the addition of 30% H2 decrease the peak flame temperature by about 19K compared to neat propane flame as clear in Fig.1. The lower of flame temperature with H2 addition is consist of the recent study of Chen and Scribano [28]. This decrease in temperature resulting from the addition of H2 can be attributed to its thermal effect. In particular, hydrogen has a higher specific heat capacity than propane[29], which requires more energy to heat the H2 -propane mixture, meaning more combustion energy is used to heat the fuel mixture itself, thus reducing the energy needed to raise the flame temperature. Furthermore, the higher laminar burning velocity of hydrogen causes the reaction zone to shift upstream towards the burner as clear in Fig.1 and 2. This shift of the reaction zone. This can enhance heat transfer from the flame and increase heat loss to the surroundings[28]. Furthermore, the higher laminar burning velocity of H2 causes the reaction zone to shift upstream towards the burner nozzle, as shown in Figures 1 and 2, resulting in a narrowing of the high-temperature zone and an increase in temperature gradients. This change in the location and shape of the reaction zone also contributes to a decrease in flame temperature, which enhances heat transfer from the flame and increases heat loss to the surrounding environment [28].
|
|
|
Fig. 1, Temperature distributions for pure propane flame (left) and propane with 30% H2 addition (right)
To isolate the chemical effect of H2 from its thermal and dilution effects, we used the approach proposed by Liu et al. [30] by introducing fictitious specie of XH2. Here assumed that this fictitious species has the same physical properties (e.g., specific heat, diffusion coefficients, gas-phase radiation coefficients, etc.) as real H2, but is not allowed to participate in any chemical reactions. In this way, the differences in results between H2 addition and XH2 can be attributed entirely to the chemical effects of H2. As can be seen in Figure 3, the addition of H2 results in a slight increase in the maximum flame temperature compared to the case of adding XH2. For instance, the maximum flame temperature increases by 5 k with the addition of XH2 compared to that of 30% H2 addition. This increase of temperature attributed to chemical effect of H2. In particular, hydrogen addition increasing the concentration of active radicals (H, O, and OH) within the flame zone. These radicals accelerate the oxidation kinetics of intermediate hydrocarbon species derived from propane, leading to a more intense and localized release of energy [10, 31].
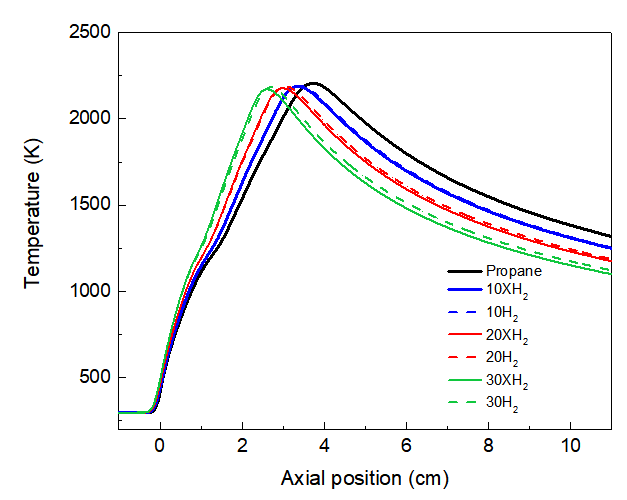
Fig. 2. Compared of temperature profiles along centerline for pure propane flame with different ration of H2/XH2 addition
3.2 Formation of benzene with H2 addition
The formation of PAHs and soot is critically governed by the chemical pathways leading to the first aromatic ring, primarily benzene (A1) [26, 27, 32]. The results presented in Figures 3 demonstrates a clear suppression of the maximum mole fraction of A1 with 30% H₂ blending. This suppression can be explained by the total effect of H₂. Moreover, it is shown in Fig. 3 that the peaks of A1 mass fraction profiles shift slightly towards the burner rim with the addition of 30%H2 as compared to neat propane, which are consistent with the change of temperature and reactions zone shown in Fig. 1.
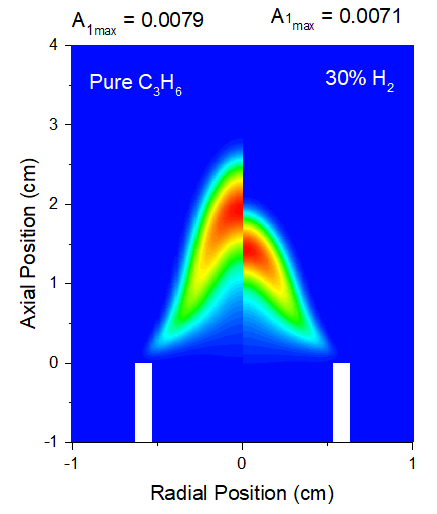
Fig. 3, A1 distributions for pure propane flame (left) and propane with 30% H2 addition (right)
The use of fictitious XH₂ species is crucial in analyzing the underlying mechanisms of the chemical effect of H2. The comparison shows that while the addition of fictitious XH₂ (simulating only the thermal and dilution effects) leads to a decrease in A1, the inhibition is more pronounced with the H₂ addition. Furthure more, Fig.4 shows that as the H2/XH2 ratio increases, the differences in results of A1 mole fraction between H2 and xH2 become more pronounced, indicating that the chemical effect increases with the addition of H2.
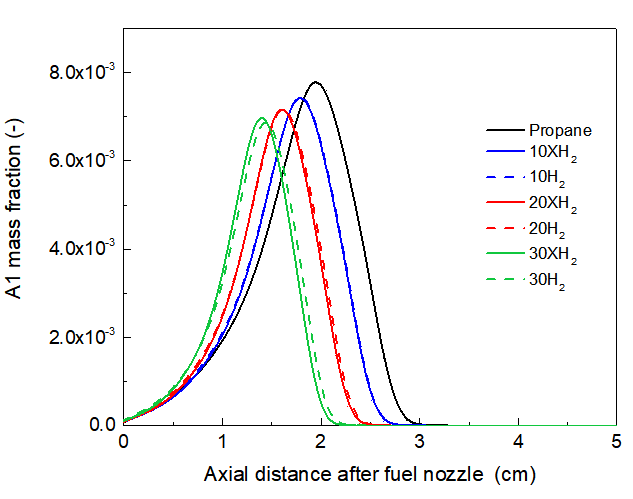
Fig. 4. Compared of A1 mole fraction along centerline for pure propane flame with different ratio of H2/XH2 addition
2.3 Formation of Small Precursors with H2 addition.
Acetylene (C₂H₂) and propargyl (C₃H₃) species are crucial for understanding the observed trends in A1. While C₂H₂ is a fundamental building block for PAH surface growth via the Hydrogen-Abstraction-C₂H₂-Addition (HACA) mechanism [27, 33, 34]. The profile of C₂H₂ is presented in Fig. 5 for comparisons among the neat propane, H2 addition, and XH2 addition flames. As mentioned before, the differences between the latter two flames reveal the chemical effect of H2. It can be seen that, although the flame temperature increases with the chemical effect of H2, the chemical effect of H2 further decreases the production of C2H2, which is consistent with the overall suppression of A1. This suggests that H₂ addition enhances the oxidative breakdown of propane fragments, reducing the pool of C₂ species available for aromatization and growth [35].
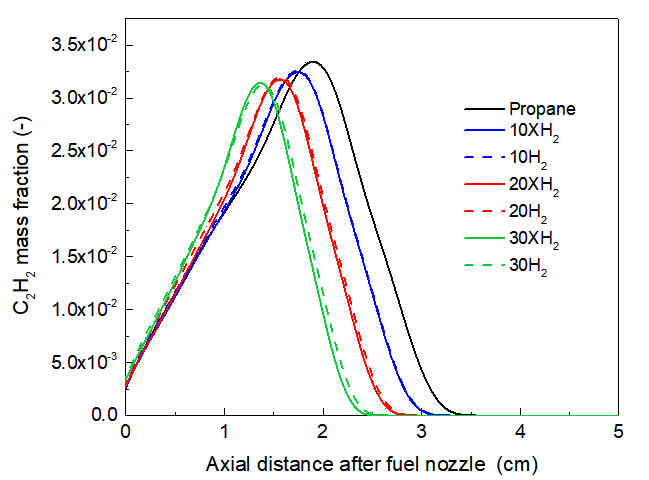
Fig. 5. Compared of C2H2 mole fraction along centerline for neat propane flame with different ration of H2/XH2 addition
The concentration of C₃H₃, a direct precursor to benzene also presented in Fig.6 as C₃H₃ radicals, a key precursor to A1 via the C₃H₃ + C₃H₃ reaction [27, 34-36]. Similarly, C₃H₃ is also suppressed as seen in Fig.6. This can be addressed from the differences between the results of XH2 and H2 flames. This inhabitation of C₃H₃ indicates that the chemical effect of hydrogen in propane flames is predominant, actively suppressing the key molecular pathways leading to the inception of soot precursors, rather than merely acting as a diluent. Furthermore, this finding aligns with the observations of Akram et al. [21] in n-dodecane flames but contrasts with studies on methane flames [5], highlighting the fuel-specific nature of H₂’s chemical effect.
Figures 3 and 4 show a clear reduction in the maximum mole fraction of A1 with 30% H₂ blending. This suppression can be explained by the chemical interaction of H₂ within the fuel stream. The increased H-atom concentration promotes the hydrogenation of small hydrocarbon radicals, potentially steering the chemical pathways away from the recombination reactions that form the first aromatic ring. For instance, propargyl (C₃H₃) radicals, a key precursor to benzene via the C₃H₃ + C₃H₃ reaction, can be scavenged by H atoms to form allene or propyne, thereby inhibiting the benzene formation route [13]. This finding aligns with the observations of Akram et al. [7] in n-dodecane flames but contrasts with studies on methane flames [19], highlighting the fuel-specific nature of the chemical role of H₂ .
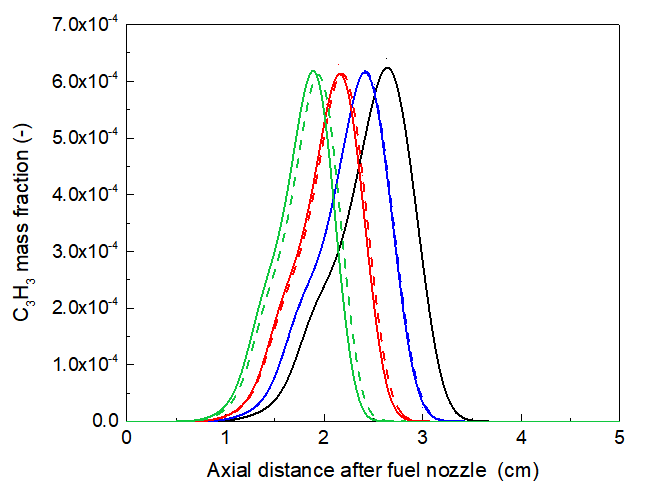
Fig. 6. Compared of C3H3 mole fraction along centerline for pure propane flame with different ration of H2/XH2 addition
Concluding Remarks
This numerical study elucidates the dual thermal/dilution and chemical effects of H2 addition on propane coflow diffusion flames. The results showed that the H2 addition lowers the peak flame temperature and narrows the high‑temperature zone due to its thermal properties and increased laminar burning velocity. More importantly, the chemical effect of H2 enhance the flame temperature due to enhances of radial species. In addition, the chemical effect of H2 suppresses the formation of A1 and its critical precursors (i.e. C₂H₂ and C₃H₃). This suppression is attributed to H2 ‑driven enhancement of radical pools, which promotes oxidative breakdown of propane fragments and inhibits reactions routes toward aromatic ring formation. The observed behavior contrasts with the results in previous studies of methane flames, highlighting the chemical role of hydrogen addition with different fuels. The results of current work provide a kinetic basis for designing cleaner combustion systems using these mixtures.
References
[1] Giurcan, V., et al., The impact of H2-enrichment on flame structure and combustion characteristic properties of premixed hydrocarbon-air flames, Fuel, 376. (2024), p. 132674, DOI No. https://doi.org/10.1016/j.fuel.2024.132674
[2] Kennedy, I.M., The health effects of combustion-generated aerosols, Proceedings of the Combustion Institute, 31. (2007), 2, pp. 2757-2770, DOI No. 10.1016/j.proci.2006.08.116
[3] Huang, R.-J., et al., High secondary aerosol contribution to particulate pollution during haze events in China, Nature, 514. (2014), 7521, pp. 218-222, DOI No. 10.1038/nature13774
[4] Zhong, W., et al., Spray-evaporation characteristics of n-pentanol/n-dodecane binary fuel at ultra-high injection pressure, Renewable Energy, 219. (2023), p. 119505, DOI No. https://doi.org/10.1016/j.renene.2023.119505
[5] Zhong, W., et al., Numerical study of spray combustion and soot emission of gasoline–biodiesel fuel under gasoline compression ignition-relevant conditions, Fuel, 310. (2022), p. 122293, DOI No. https://doi.org/10.1016/j.fuel.2021.122293
[6] Zhong, W., et al., Experimental study on in-flame soot formation and soot emission characteristics of gasoline/hydrogenated catalytic biodiesel blends, Fuel, 289. (2021), p. 119813, DOI No. https://doi.org/10.1016/j.fuel.2020.119813
[7] Zhong, W., et al., Experimental and modeling study of the autoignition characteristics of gasoline/hydrogenated catalytic biodiesel blends over low-to-intermediate temperature, Fuel, 313. (2022), p. 122919, DOI No. https://doi.org/10.1016/j.fuel.2021.122919
[8] Zang, G., et al., H2 production through natural gas reforming and carbon capture: A techno-economic and life cycle analysis comparison, International Journal of Hydrogen Energy, 49. (2024), pp. 1288-1303
[9] Małek, A., Low-emission hydrogen for transport—A technology overview from hydrogen production to its use to power vehicles, Energies, 18. (2025), 16, p. 4425
[10] Wang, Y., et al., Effects of hydrogen blending on combustion and pollutant emission of propane/air in a model furnace with a rotary kiln burner, Thermal Science and Engineering Progress, 47. (2024), p. 102330, DOI No. https://doi.org/10.1016/j.tsep.2023.102330
[11] Jowkar, S., et al., Hydrogen-enriched propane combustion in a lean premixed burner: LES study on flashback, emissions, and combustion instability, Fuel, 381. (2025), p. 133377, DOI No. https://doi.org/10.1016/j.fuel.2024.133377
[12] Effects of hydrogen and steam addition on laminar burning velocity of methane–air premixed flame: experimental and numerical analysis %J International journal of hydrogen energy, 37. (2012), 11, pp. 9412-9422
[13] Al-Hamamre, Z.,J.J.I.j.o.h.e. Yamin, The effect of hydrogen addition on premixed laminar acetylene–hydrogen–air and ethanol–hydrogen–air flames, 38. (2013), 18, pp. 7499-7509
[14] Arslan, E.,N.J.I.J.o.H.E. Kahraman, The effects of hydrogen enriched natural gas under different engine loads in a diesel engine, 47. (2022), 24, pp. 12410-12420
[15] Benaissa, S., et al., Effect of hydrogen addition on the combustion characteristics of premixed biogas/hydrogen-air mixtures, 46. (2021), 35, pp. 18661-18677
[16] Cheng, Y., et al., Kinetic analysis of H2 addition effect on the laminar flame parameters of the C1–C4 n-alkane-air mixtures: From one step overall assumption to detailed reaction mechanism, 40. (2015), 1, pp. 703-718
[17] De Iuliis, S., et al., Effect of hydrogen addition on soot formation in an ethylene/air premixed flame, 106. (2012), 3, pp. 707-715
[18] Ezenwajiaku, C., et al., Investigation of the effect of hydrogen addition on soot and PAH formation in ethylene inverse diffusion flames by combined LII and PAH LIF, 361. (2024), p. 130613
[19] Liu, F., et al., Effect of hydrogen and helium addition to fuel on soot formation in an axisymmetric coflow laminar methane/air diffusion flame, International Journal of Hydrogen Energy, 39. (2014), 8, pp. 3936-3946, DOI No. https://doi.org/10.1016/j.ijhydene.2013.12.151
[20] Sun, Z., et al., Effects of hydrogen and nitrogen on soot volume fraction, primary particle diameter and temperature in laminar ethylene/air diffusion flames, Combustion and Flame, 175. (2017), pp. 270-282, DOI No. https://doi.org/10.1016/j.combustflame.2016.08.031
[21] Akram, M.Z., Study of hydrogen impact on lean flammability limit and burning characteristics of a kerosene surrogate, Energy, 231. (2021), p. 120925, DOI No. https://doi.org/10.1016/j.energy.2021.120925
[22] Kashir, B.,S. Tabejamaat, A numerical study on the effects of H2 addition in non-premixed turbulent combustion of C3H8–H2–N2 mixture using a steady flamelet approach, International Journal of Hydrogen Energy, 38. (2013), 23, pp. 9918-9927, DOI No. https://doi.org/10.1016/j.ijhydene.2013.05.103
[23] ‘***’, http://www.ansys.com.
[24] Wang, H.,M. Frenklach, A detailed kinetic modeling study of aromatics formation in laminar premixed acetylene and ethylene flames, Combustion and Flame, 110. (1997), 1, pp. 173-221, DOI No. https://doi.org/10.1016/S0010-2180(97)00068-0
[25] M Mahmoud, N.,B. M Ahmed, Computational study of small-scale laminar coflow diffusion flames: influences of fuel dilution on the negative buoyant flame, IOP Conference Series: Earth and Environmental Science, 146. (2018), p. 012021, DOI No. 10.1088/1755-1315/146/1/012021
[26] Mahmoud, N.M., et al., Effects of fuel inlet boundary condition on aromatic species formation in coflow diffusion flames, Journal of the Energy Institute, 92. (2019), 2, pp. 288-297, DOI No. https://doi.org/10.1016/j.joei.2018.01.007
[27] Mahmoud, N.M., et al., Flame Structure and Soot-Precursor Formation of Coflow n-Heptane Diffusion Flame Burning in O2/N2 and O2/CO2 Atmosphere, Journal of Energy Engineering, 147. (2021), 4, p. 04021027, DOI No. doi:10.1061/(ASCE)EY.1943-7897.0000776
[28] Cheng, X.,G. Scribano, Effects of hydrogen addition on the laminar premixed flames and emissions of methane and propane, International Journal of Hydrogen Energy, 53. (2024), pp. 1-16, DOI No. https://doi.org/10.1016/j.ijhydene.2023.12.040
[29] You, C., et al., Effect of N2/CO2 Dilution Ratios on Explosion Characteristics of Hydrogen/Propane Mixtures, ACS omega, 10. (2025), 8, pp. 7989-7998
[30] Liu, F., et al., The Chemical Effects of Carbon Dioxide as an Additive in an Ethylene Diffusion Flame: Implications for Soot and NOx Formation, combustion and flame, 125. (2001), pp. 778-787
[31] Li, Y., et al., Effect of initial pressure on hydrogen/propane/air flames in a closed duct, International Journal of Hydrogen Energy, 64. (2024), pp. 947-957, DOI No. https://doi.org/10.1016/j.ijhydene.2024.03.352
[32] Mahmoud, N.M., et al., Chemical Effects of CO2 and H2O Addition on Aromatic Species in Ethanol/Air Diffusion Flame, Combustion Science and Technology. (2020), pp. 1-19, DOI No. 10.1080/00102202.2020.1776705
[33] Mahmoud, N.M., et al., Impact of n-butanol addition to hydrogenated catalytic biodiesel fueled a constant volume combustion chamber; a computational study, Energy Sources, Part A: Recovery, Utilization, and Environmental Effects, 45. (2023), 4, pp. 12553-12569, DOI No. 10.1080/15567036.2023.2273986
[34] Mahmoud, N.M., et al., On the effect of injection pressure on spray combustion and soot formation processes of gasoline/second generation biodiesel blend, Thermal Science, 28. (2024), 00, pp. 3967-3978
[35] Frenklach, M., Reaction mechanism of soot formation in flames, Physical Chemistry Chemical Physics, 4. (2002),
[36] Mahmoud, N.M., et al., Coupled Effects of Carbon Dioxide and Water Vapor Addition on Soot Formation in Ethylene Diffusion Flames, Energy & Fuels, 33. (2019), 6, pp. 5582-5596, DOI No. 10.1021/acs.energyfuels.9b00192