Pulmonary Toxicity and Mild Cardiac Alterations Induced by Datura stramonium Extract in Swiss Albino Mice
السمّية الرئوية والتغيرات القلبية الطفيفة الناتجة عن مستخلص نبات الداتورا (Datura stramonium) في الفئران البيضاء السويسرية
Raja Abdullah Aljibali¹, Abdelmuhsen M. Abusneina², Ali Mohammad Kasheesh¹, Soad Mohamed Alwirfli¹*
¹ Department of Forensic Sciences, Faculty of Biomedical Sciences, University of Benghazi, Libya
² Department of Molecular Diagnostics, Faculty of Biomedical Sciences, University of Benghazi, Libya
Corresponding author: Soad Mohamed Alwirfli — soad.mohammed@uob.edu.ly
DOI: https://doi.org/10.53796/hnsj610/8
Arabic Scientific Research Identifier: https://arsri.org/10000/610/8
Volume (6) Issue (10). Pages: 103 - 112
Received at: 2025-09-07 | Accepted at: 2025-09-15 | Published at: 2025-10-01
Abstract: Background: Datura stramonium (jimsonweed) contains tropane alkaloids (atropine, scopolamine, hyoscyamine) known for their anticholinergic and systemic toxicities. While hepatic, renal, and reproductive effects are well documented, its cardiopulmonary toxicity remains underexplored. Methods: Male Swiss albino rodents were administered aqueous leaf extract of D. stramonium via acute oral doses (12.5–200 mg/kg) and sub-chronic weekly doses (0.36, 0.7, 4 mg/kg) for 28–40 days. Groups included control, low, medium, high dose, and recovery cohorts. Body weight, relative heart and lung weights, and histopathology (H&E) of heart and lung tissues were assessed. Results: No mortality or significant changes in body or organ weights were observed, however, the high-dose groups showed an upward trends in lung weights. Cardiac histology was largely preserved, with mild alterations, such as focal vacuolation or myofibrillar degeneration, observed at higher doses. Pulmonary tissues exhibited dose-dependent lesions, including alveolar congestion, septal thickening, bronchiolar epithelial detachment, hemorrhage, and inflammatory infiltration. Recovery groups showed partial histological improvement. Conclusion: D. stramonium leaf extract induces dose-related pulmonary toxicity with minimal structural cardiotoxicity under the studied conditions. These findings highlight the lungs as particularly susceptible targets and reinforce the plant's multi-organ toxicological profile. Functional studies and public health awareness are warranted.
Keywords: Datura stramonium, tropane alkaloids, pulmonary toxicity, cardiotoxicity, histopathology, Swiss albino mice.
المستخلص: الخلفية: يحتوي نبات الداتورا (Datura stramonium) المعروف أيضًا بعشبة جيمسون على قلويدات التروبان (الأتروبين، السكوبولامين، الهيوسيامين) التي تُعرف بخصائصها المضادة للكولين وسمّيتها الجهازية. وبينما تم توثيق تأثيراته الكبدية والكلوية والتناسلية بشكل جيد، فإن سُميته القلبية-الرئوية ما زالت غير مستكشفة بالشكل الكافي. الطرق: تم إعطاء قوارض بيضاء سويسرية من الذكور مستخلصًا مائيًا من أوراق الداتورا عن طريق جرعات فموية حادة (12.5–200 ملغ/كغ) وجرعات تحت-مزمنة أسبوعية (0.36، 0.7، 4 ملغ/كغ) لمدة 28–40 يومًا. شملت المجموعات: مجموعة ضابطة، جرعة منخفضة، متوسطة، عالية، ومجموعة تعافي. جرى تقييم وزن الجسم، والأوزان النسبية للقلب والرئتين، إضافةً إلى الفحص النسيجي المرضي (H&E) لأنسجة القلب والرئة. النتائج: لم يُسجَّل أي نفوق أو تغيّرات كبيرة في أوزان الجسم أو الأعضاء، غير أن مجموعات الجرعات العالية أظهرت اتجاهًا تصاعديًا في أوزان الرئتين. حافظ النسيج القلبي إلى حد كبير على سلامته، مع ظهور تغيّرات طفيفة مثل التفرغ البؤري أو التنكس الليفي العضلي عند الجرعات المرتفعة. أظهرت أنسجة الرئة إصابات مرتبطة بالجرعة، شملت احتقانًا حُوَيْصِليًا، وزيادة في سُمك الحواجز، وانفصال الظهارة القصيبية، ونزفًا، وتسللًا التهابيًا. أما مجموعات التعافي فقد أظهرت تحسنًا نسيجيًا جزئيًا. الاستنتاج: يُحدث مستخلص أوراق الداتورا سمية رئوية مرتبطة بالجرعة مع سمية قلبية بنيوية طفيفة ضمن الظروف المدروسة. وتُبرز هذه النتائج الرئتين كأهداف أكثر عرضة للتأثر، كما تؤكد على الملف السُمّي متعدّد الأعضاء للنبات. ويوصى بإجراء دراسات وظيفية إضافية وزيادة التوعية الصحية العامة.
الكلمات المفتاحية: الداتورا (Datura stramonium)، قلويدات التروبان، السمية الرئوية، السمية القلبية، علم الأمراض النسيجي (الهستوباثولوجي)، الفئران البيضاء السويسرية.
Datura stramonium L. (Solanaceae), commonly referred to as jimsonweed or thorn apple, is a widely distributed plant recognized for its dual role in traditional medicine and toxicology. Its pharmacological and toxic properties are attributed to its rich content of tropane alkaloids—primarily atropine, scopolamine, and hyoscyamine—which act as competitive antagonists at muscarinic acetylcholine receptors. This antagonism produces a classical anticholinergic toxidrome, characterized by symptoms including mydriasis, xerostomia, tachycardia, hyperthermia, hallucinations, urinary retention, and, in severe cases, seizures, coma, or death (Krenzelok, 2010; Shifa et al., 2024). Earlier reports also documented clusters of adolescent and young adult intoxications, underscoring the plant’s persistent role in recreational abuse (Klein-Schwartz & Odera, 1984).
Ethnobotanical records document the historical use of D. stramonium in Africa, Asia, and the Americas for the treatment of asthma, cough, and pain (Al-Gadi, 1997; Burkill, 2000). In southern Africa, ethnobotanical surveys confirm that D. stramonium remains both a medicinal and hallucinogenic plant, widely recognized for its dual therapeutic and toxic potential (Setshogo, 2015). Traditional preparations, such as infusions or smoked formulations, were administered for their bronchodilatory and analgesic effects. However, the narrow therapeutic index of the plant renders it prone to accidental overdose, particularly in pediatric cases, as well as misuse in recreational or ritual contexts (Boumba et al., 2004; Onen et al., 2002; Al-Shaikh & Sablay, 2005). Recent reviews have highlighted the rising incidence of recreational misuse and intoxication from D. stramonium, emphasizing its growing impact on public health and the need for improved toxicological awareness (Aćimović, 2025; Lorrai et al., 2024). Forensic studies have frequently identified pulmonary congestion and cerebral edema as consistent findings in fatal intoxications (Le Garff et al., 2016).
Phytochemical studies have shown that the alkaloid content and atropine-to-scopolamine ratios in D. stramonium can vary considerably based on species, plant part, geographical origin, and environmental conditions (Berkov & Zayed, 2004; Sharma et al., 2021). Analytical confirmation of exposure in clinical and forensic settings is typically achieved through GC–MS or LC–MS/MS analysis of biological fluids (Nogué et al., 1995; Steenkamp et al., 2004).
A growing body of experimental evidence confirms the multi-organ toxicity of D. stramonium. Studies in rodents have demonstrated hepatic necrosis, renal tubular degeneration, and testicular injury following exposure to both aqueous and alcoholic extracts (Gidado et al., 2007; Ogunmoyole et al., 2019; Pretorius & Marx, 2006). Our previous work has also shown significant hepatotoxic, nephrotoxic, and reproductive effects in Swiss albino mice exposed to aqueous leaf extracts of the plant.
Despite clinical reports of cardiac arrhythmias and respiratory complications in acute intoxications (Krenzelok, 2010; Codina et al., 2025; Shifa et al., 2024), controlled experimental studies on cardiopulmonary toxicity remain limited. Functional disturbances such as tachycardia may occur without detectable structural changes, particularly in the heart (Krenzelok, 2010; Codina et al., 2025). In contrast, the lungs, due to their extensive vascular surface area and direct exposure to circulating toxins, may represent a primary target for early structural injury, particularly under conditions of oxidative or inflammatory stress (Sharma et al., 2021; Pretorius & Marx, 2006).
The present study aimed to evaluate the acute and sub-chronic effects of an aqueous Datura stramonium leaf extract on the cardiac and pulmonary tissues of Swiss albino mice. Parameters assessed included clinical observations, body weight trends, relative organ weight indices, and histopathological changes in the heart and lungs. Findings were interpreted in the context of prior research on systemic organ toxicity, including our earlier demonstrations of testicular degeneration (Alwirfli et al., 2021a) and hepatic and renal injury (Alwirfli et al., 2021b), to determine whether these critical organs exhibit dose-dependent structural alterations following exposure.
Ethical Approval and Animal Welfare
All experimental procedures were conducted in accordance with the ethical guidelines of the Institutional Animal Ethics Committee, Faculty of Biomedical Sciences, University of Benghazi, Libya (Approval No. BMS/ETH/2025/01). Care and handling of animals adhered to international standards for the humane treatment of laboratory animals.
Experimental Animals
Adult male Swiss albino mice (25–30 g, aged 8–10 weeks) were obtained from the university’s animal facility. Animals were housed in standard polypropylene cages under controlled environmental conditions (temperature: 23 ± 2°C; relative humidity: 50–60%; light/dark cycle: 12 h/12 h), with free access to commercial food and water. Animals were allowed to acclimatize for one week prior to treatment.
Plant Material and Extract Preparation
Fresh leaves of Datura stramonium were collected from peri-urban areas of Benghazi during the peak flowering season. The plant was taxonomically authenticated by a botanist at the Department of Botany, Faculty of Science, University of Benghazi. The leaves were washed, shade-dried, and pulverized into a fine powder. The aqueous extract was prepared by macerating the powder in distilled water (1:10 w/v) for 48–72 hours with intermittent shaking. The extract was filtered and concentrated under reduced pressure at ≤40 °C using a rotary evaporator, yielding a semi-solid residue. The extract was stored at 4 °C and freshly reconstituted in distilled water before administration.
Acute Toxicity Study
A total of 16 mice were randomly assigned to six groups (n = 2–3 per group). Animals received a single oral dose of the extract at 12.5, 25, 50, 100, or 200 mg/kg, or vehicle (distilled water) as a control. Mice were monitored continuously for the first 24 hours and then daily for a total of 96 hours. Observations included mortality, locomotion, grooming, piloerection, respiratory rate, and general behavior.
Sub-Chronic Toxicity Study
Thirty-six mice were randomly divided into four groups (n = 9 per group):
- Group I (Control): Distilled water
- Group II (Low Dose): 0.36 mg/kg
- Group III (Medium Dose): 0.7 mg/kg
- Group IV (High Dose): 4 mg/kg
The extract was administered orally via gavage once weekly for 40 days. Body weights were recorded weekly. Mice were observed daily for clinical signs, including food intake, activity levels, and signs of discomfort or toxicity.
Organ Collection and Weight Analysis
At the end of the study, animals were anesthetized using light ether and euthanized via cervical dislocation. The heart and lungs were carefully excised, rinsed in cold normal saline, blotted dry, and weighed. Relative organ weights were calculated using the formula:
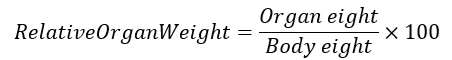
Histopathological Examination
Samples from the left ventricle of the heart and both lobes of the lungs were fixed in 10% neutral-buffered formalin for at least 24 hours. Ti were dehydrated in a graded series of alcohols, cleared in xylene, embedded in paraffin wax, and sectioned at a thickness of 5 µm using a rotary microtome. Sections were stained with hematoxylin and eosin (H&E) and examined under a light microscope at magnifications of ×100 to ×400. Representative photomicrographs were captured. Lesions were graded semi-quantitatively as mild, moderate, or severe.
Statistical Analysis
Data are presented as mean ± standard deviation (SD). Statistical comparisons among groups were conducted using one-way analysis of variance (ANOVA) followed by Tukey’s post hoc test for multiple comparisons. Where assumptions of normality were violated, non-parametric alternatives were considered. A p-value < 0.05 was considered statistically significant. All analyses were performed using SPSS version 25.0 (IBM Corp., Armonk, NY, USA).
The administration of aqueous Datura stramonium leaf extract produced a series of dose-related physiological and histological responses, most notably affecting pulmonary tissue. Across both the acute and sub-chronic exposure protocols, no mortality was observed in any group, indicating that the extract, at the tested doses, was not lethally toxic. However, subtle clinical signs—such as transient reductions in spontaneous activity and grooming—were noted within hours post-dosing in some treated animals, particularly at higher doses. These behaviors resolved spontaneously within the same day, and no neurological signs such as tremors, seizures, or paralysis were recorded. Over the 40-day exposure period, all animals exhibited a progressive increase in body weight, with no statistically significant differences observed between the control and treated groups. This suggests that the extract did not substantially impair overall metabolism or feeding behavior. Despite this, a downward trend in body weight gain was noted in the high-dose group, hinting at possible subclinical systemic stress.
Upon euthanasia, organ weights were recorded to assess potential changes in organ mass as a reflection of underlying pathology. Relative heart weights remained comparable across all groups, and statistical analysis revealed no significant differences between them. A similar pattern was observed in relative lung weights, although slightly elevated values were recorded in the low- and mid-dose groups. These variations were not statistically significant, but they may inform future studies with larger sample sizes or extended durations (Figure 1).
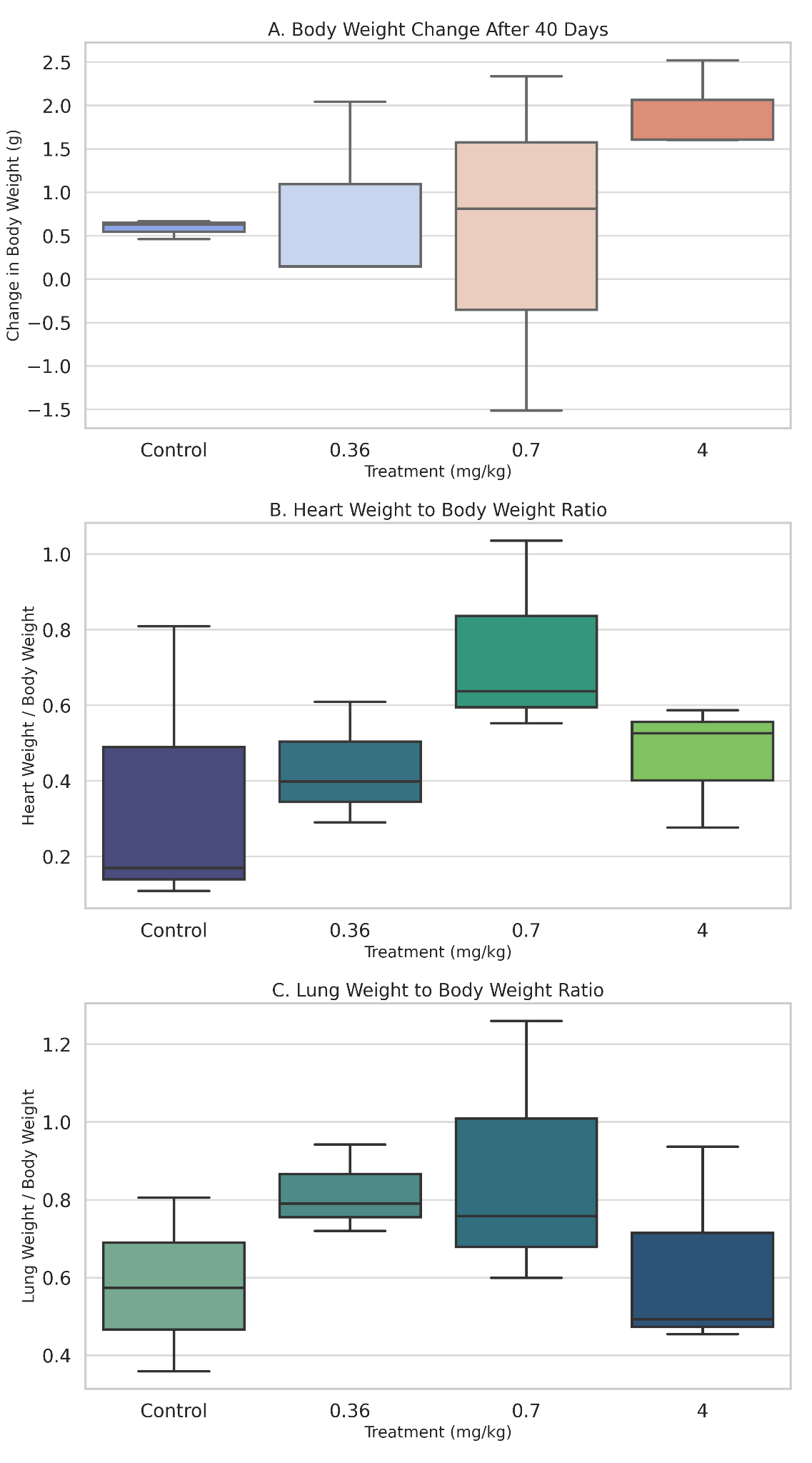
Figure 1. Showing (A) body weight change, (B) relative heart weight, and (C) relative lung weight of mice treated with Datura stramonium extract for 40 days. Individual data points are shown (n = 3 per group). No significant differences were observed.
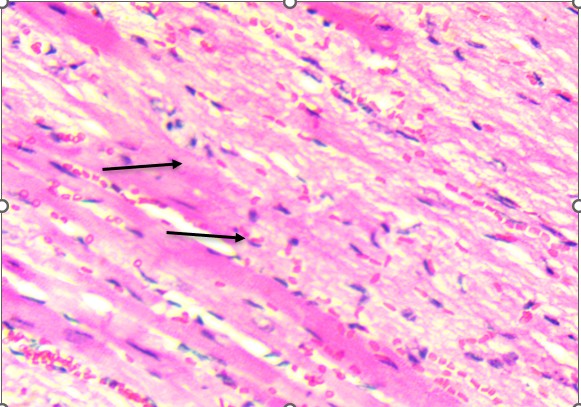
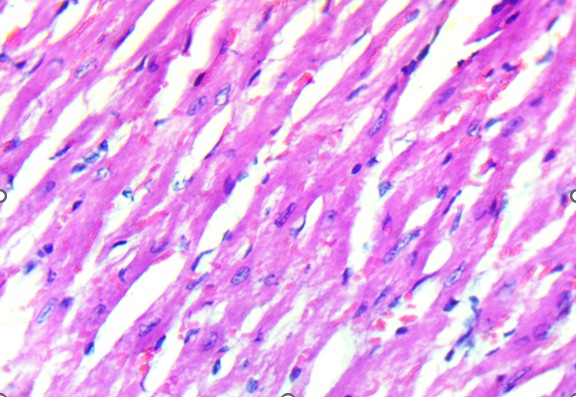
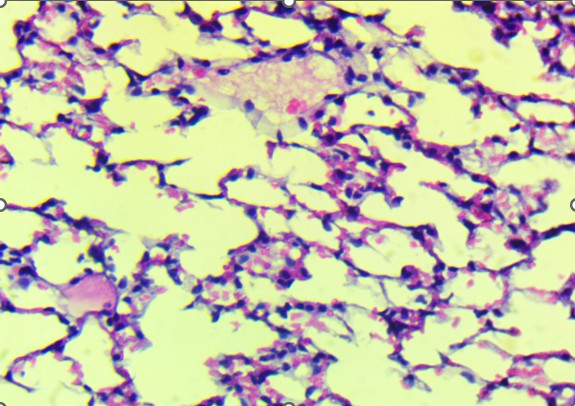
Histological analysis revealed that the heart was structurally preserved in all groups. In both control and treated animals, the cardiac tissue exhibited well-aligned myocardial fibers with distinct cross-striations and centrally located nuclei. No evidence of myocardial necrosis, interstitial edema, vascular congestion, or inflammatory infiltration was observed. Even in high-dose groups, the myocardium retained normal architecture, supporting the conclusion that the extract did not induce structural cardiotoxicity under these experimental conditions (Figure 2). In contrast, the lungs demonstrated a more apparent dose-dependent vulnerability to the extract. Control animals exhibited healthy pulmonary architecture, with well-expanded alveoli, thin interalveolar septa, and intact bronchiolar epithelium. These features were largely maintained in the low-dose group, although a few animals showed mild epithelial irregularities.
|
|
|
A. Control. . B. Treated
Figure 2. Heart of Swiss albino mice: (A) Control myocardium showing intact fibers with clear striations and central nuclei. (B) Treated myocardium displaying preserved architecture without necrosis, edema, or inflammatory infiltrates.
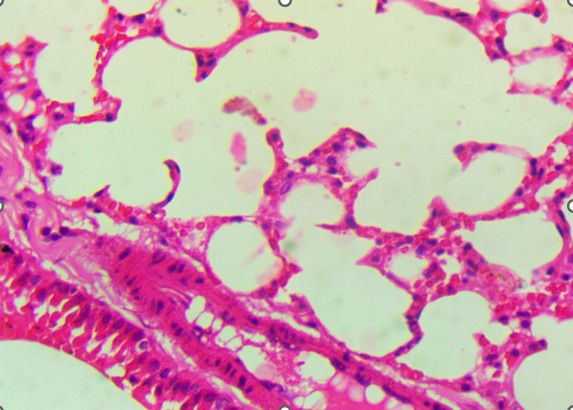
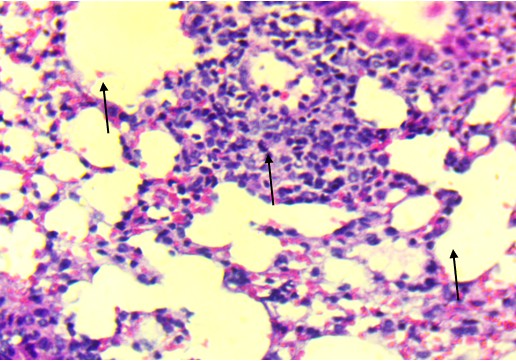
In the mid-dose group (0.7 mg/kg), histopathological changes became more apparent. The alveolar septa were mildly thickened, and there was evidence of focal inflammatory infiltration, particularly in the peribronchiolar regions. These findings suggest early-stage toxic alveolitis, possibly mediated by oxidative or inflammatory mechanisms triggered by the plant’s alkaloids. The high-dose group (4 mg/kg) revealed the most prominent pulmonary lesions.
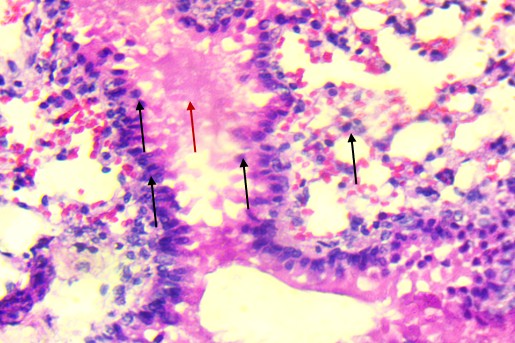
Bronchiolar epithelium appeared partially detached, and several lung sections displayed eosinophilic proteinaceous material within the airway lumens. Alveolar spaces were noticeably reduced, and focal hemorrhages were present, indicating increased capillary permeability or endothelial injury (Figure 3).
|
A. Control: Normal alveoli and septa. |
B. T1: Comparable to control with minimal epithelial irregularities. |
|
C. T2: Septal thickening and peribronchiolar infiltration. |
D. T3 epithelial detachment. |
|
E. T3 intra-alveolar hemorrhage. |
F. Representative treated: Multifocal hemorrhage and inflammation. |
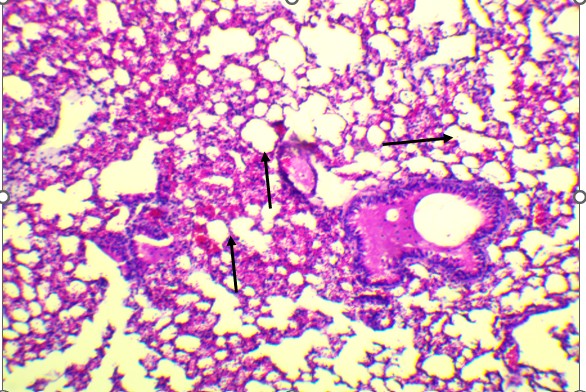
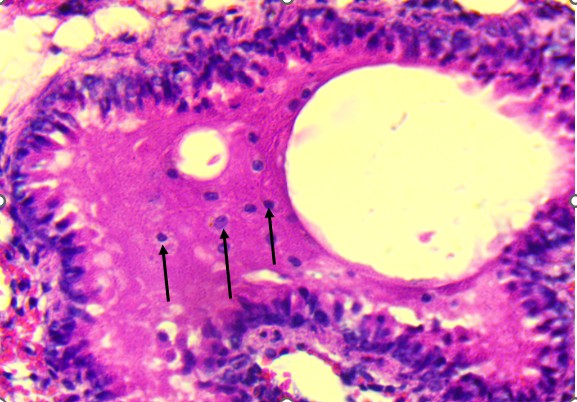
Figure 3. Lungs of Swiss albino mice: (A) Control with intact alveoli, (B) T1 minimal changes, (C) T2 septal thickening, (D) T3 epithelial detachment, (E) T3 intra-alveolar hemorrhage, (F) Representative treated lung with multifocal hemorrhage.
Despite the severity of these findings, there was no widespread consolidation or fibrotic remodeling, implying that the damage remained in the acute, potentially reversible phase. Animals from the recovery group that received the high dose followed by a treatment-free period showed partial resolution of pulmonary lesions. While some inflammatory features had regressed, mild septal thickening and epithelial irregularities persisted, indicating incomplete histological recovery within the observation window.
These histological observations were consistent with the recorded trends in organ weights, although the functional impact of these structural changes remains to be explored in future studies. The heart, while frequently implicated in clinical reports of Datura-related toxicity (e.g., arrhythmias), appeared resistant to structural injury in this model. Conversely, the lungs exhibited mild but consistent dose-dependent damage, highlighting their greater vulnerability to systemic or direct toxic effects of Datura stramonium alkaloids.
The present study investigated the cardiopulmonary effects of aqueous Datura stramonium leaf extract in male Swiss albino mice, revealing a consistent pattern of organ-selective toxicity. Despite repeated exposure across 40 days, animals tolerated the extract well, exhibiting no mortality and maintaining normal feeding behavior and progressive weight gain throughout the experiment. Clinical observations were limited to transient reductions in spontaneous activity and grooming in higher-dose groups, which resolved spontaneously within hours. The absence of significant weight loss or behavioral deterioration suggested that the administered doses did not compromise systemic homeostasis or induce overt toxicity.
As shown in Figure 1A, body weight changes remained within normal ranges across all groups, including those receiving the highest dose of extract. Likewise, relative heart and lung weights (Figures 1B and 1C) did not differ significantly between control and treatment groups, though mild upward trends were noted at mid and high doses. These findings suggest that gross organ morphology remained essentially unchanged, underscoring the value of histological analysis in identifying subtler tissue-specific changes that may precede measurable alterations in organ mass.
Histological examination of the heart revealed well-preserved myocardial architecture in both control and treated animals (Figure 2A–B). Myofibrils were regularly aligned, cross-striations were intact, and nuclei were centrally placed, findings consistent with the absence of cellular injury or inflammatory infiltration. Even at the highest extract dose, the myocardium exhibited no signs of necrosis, edema, or vascular compromise, indicating that D. stramonium did not induce structural cardiotoxicity under these experimental conditions. This aligns with clinical observations of Datura poisoning in humans, where tachycardia and arrhythmias are common yet typically reversible, reflecting autonomic dysregulation rather than direct myocardial damage (Krenzelok, 2010; Al Shaikh & Sablay, 2005).
In contrast to the heart, the lungs exhibited apparent dose-related histological alterations (Figure 3). In control animals (Figure 3A), pulmonary tissue appeared intact with open alveolar spaces, thin interalveolar septa, and unremarkable bronchiolar epithelium. This structure was mainly preserved in the low-dose group (Figure 3B), with only minor irregularities. However, lungs from the mid-dose group (Figure 3C) began to show mild septal thickening and peribronchiolar inflammatory infiltration, indicating the onset of localized alveolar injury.
At the highest dose (Figures 3D and 3E), the lung tissue revealed more severe pathology, including bronchiolar epithelial detachment, eosinophilic deposits, reduced alveolar spaces, and focal hemorrhage. These findings suggest a toxic alveolitis-like response, potentially driven by a combination of muscarinic receptor blockade, which can impair airway regulation and mucosal secretions, and oxidative stress, which may provoke capillary leakage and inflammatory activation. Similar histological patterns have been reported in experimental and forensic studies of acute Datura intoxication, often correlating with respiratory distress or pulmonary edema observed at autopsy (Boumba et al., 2004; Le Garff et al., 2016; Shifa et al., 2024).
Of particular interest, lungs from recovery animals (Figure 3F) showed partial resolution of lesions. Although inflammation was reduced and some epithelial reorganization had occurred, residual septal thickening persisted, indicating that while some of the extract’s pulmonary effects are reversible, complete histological recovery may require a more extended withdrawal period or additional therapeutic support. This partial reversibility aligns with clinical cases in which respiratory symptoms improve following cessation of exposure and supportive care.
These cardiopulmonary findings contribute to the growing body of literature describing the multi-organ toxicity profile of Datura stramonium. Previous work has documented hepatic, renal, and reproductive toxicity in both experimental and clinical contexts (Gidado et al., 2007; Ogunmoyole et al., 2019; Pretorius & Marx, 2006). When viewed alongside these earlier results, the current study reinforces the concept of organ-selective susceptibility, with the lungs appearing particularly sensitive, potentially due to their extensive vascularization and proximity to circulating toxic metabolites.
Mechanistically, the observed pulmonary alterations are likely attributable to the tropane alkaloids in D. stramonium, notably atropine and scopolamine. These compounds antagonize muscarinic receptors, which are abundant in pulmonary tissue and involved in regulating airway tone and secretion. At high doses, this disruption may contribute to bronchial constriction, epithelial stress, and increased vascular permeability, all of which could lead to the histological changes observed in this study. Additionally, oxidative and inflammatory pathways may be triggered by other alkaloids or phytochemicals in the extract, further exacerbating tissue damage (Sharma et al., 2021; Louie et al., 2003).
These findings extend earlier reports of organ-selective toxicity of Datura stramonium. Previous studies have shown that aqueous leaf extracts induce marked testicular degeneration, characterized by disorganized seminiferous tubules and impaired spermatogenesis (Alwirfli et al., 2021a), as well as significant hepatic and renal pathology, including hepatocellular necrosis and tubular degeneration (Alwirfli et al., 2021b). Together with the present observations of pulmonary vulnerability, these results strengthen the concept of multi-organ susceptibility, where specific tissues, such as the liver, kidney, reproductive organs, and lung, exhibit greater structural sensitivity than the heart. This organ-selective pattern may reflect differences in tissue perfusion, metabolic activation, or antioxidant defense capacity. While no significant changes in organ weight or gross behavior were evident, microscopic tissue injury was clearly detectable, particularly in the lungs. This suggests that functional assessments—such as pulmonary function testing, ECG analysis, echocardiography, and biomarker assays (e.g., troponin, cytokines)—could reveal additional subclinical effects not captured by histology alone. Furthermore, quantifying the alkaloid content of the extract, conducting longer-term recovery assessments, and including female subjects in future experiments will deepen our understanding of both exposure risks and recovery dynamics.
Aqueous Datura stramonium extract induces significant, dose-dependent pulmonary toxicity, marked by alveolar congestion, septal thickening, epithelial detachment, hemorrhage, and inflammatory infiltration. In contrast, cardiac alterations were mild, limited to interstitial edema without structural necrosis. These findings highlight the lungs as the primary target organ, emphasize the need for pulmonary monitoring in poisoning cases, and expand current understanding of Datura‘s multi-organ toxicity profile.
The authors declare no conflict of interest.
Aćimović, M. (2025). Datura stramonium – A dangerous weed and alternative drug of abuse: An overview of poisoning cases in the 21st century. Planta Medica, 91, 353–370.
Al‑Gadi, A. (1997). Usage of some plants in Libyan folk‑medicine. Al‑Fateh University Press.
Al‑Shaikh, A., & Sablay, Z. (2005). Hallucinogenic plant poisoning in children. Saudi Medical Journal, 26, 118–121.
Alwirfli, S. M., Elwarfali, S. M., Aljibali, R. A., & Abusneina, A. M. (2021a). Datura stramonium leaf extract toxic effects on testis in Swiss albino mice. Scientific Journal for the Faculty of Science – Sirte University, 1(1), 58–67.
Alwirfli, S. M., Elwarfali, S. M., Aljibali, R. A., & Abusneina, A. M. (2021b). The toxic effects of Datura stramonium leaf extract to liver and kidney in Swiss albino mice. Benghazi Modern University Journal, August, 33–45.
Berkov, S., & Zayed, R. (2004). Comparison of tropane alkaloid spectra between Datura innoxia grown in Egypt and Bulgaria. Zeitschrift für Naturforschung C, 59, 184–186.
Boumba, V., Mitselou, A., & Vougiouklakis, T. (2004). Fatal poisoning from ingestion of Datura stramonium seeds. Veterinary and Human Toxicology, 46, 81–82.
Burkill, H. M. (2000). The Useful Plants of West Tropical Africa. Vol. 5: Families S–Z, Addenda. Royal Botanic Gardens, Kew.
Codina, P., Tomasoni, D., Aimo, A., & Lee, M. (2025). What’s new heart failure? February 2025. European Journal of Heart Failure, 27(2), 185-189.
Gidado, A., Zainab, A., Hadiza, M., Serah, D., Anas, H., & Milala, M. (2007). Toxicity studies of ethanol extract of leaves of Datura stramonium in rats. African Journal of Biotechnology, 6, 1012–1015.
Klein‑Schwartz, W., & Odera, G. (1984). Jimson weed intoxication in adolescents and young adults. American Journal of Diseases of Children, 138, 737–739.
Krenzelok, E. P. (2010). Aspects of Datura poisoning and treatment. Clinical Toxicology, 48, 104–110.
Le Garff, E., Mesli, V., & Raul, J. S. (2016). Fatal Datura stramonium poisoning with pulmonary involvement: Autopsy case report. Forensic Science International, 266, e1–e5.
Lorrai, R., Cavaterra, D., Giammaria, S., Sbardella, D., Tundo, G. R., & Boccaccini, A. (2024). Eye diseases: When the solution comes from plant alkaloids. Planta Medica, 90, 426–439.
Louie, M., Kondor, N., & Dewitt, J. (2003). Gene expression in cadmium‑tolerant Datura innoxia: Detection and characterization of cDNAs induced in response to cadmium. Plant Molecular Biology, 52, 81–89.
Nogué, S., Pujol, L., Sanz, P., & de la Torre, R. (1995). Datura stramonium poisoning: Identification of tropane alkaloids in urine by GC–MS. Journal of International Medical Research, 23, 132–137.
Ogunmoyole, T., Adeyeye, R. I., Olatilu, B. O., Akande, O. A., & Agunbiade, O. J. (2019). Multiple organ toxicity of Datura stramonium seed extracts. Toxicology Reports, 6, 983–989.
Onen, C., Othol, D., Mbwana, S., & Manuel, I. (2002). Datura stramonium mass poisoning in Botswana. South African Medical Journal, 92, 213–214.
Pretorius, E., & Marx, J. (2006). Datura stramonium treatment and possible effects on prenatal development. Environmental Toxicology and Pharmacology, 21, 331–337.
Setshogo, M. P. (2015). A review of some medicinal and/or hallucinogenic Solanaceous plants of Botswana: The genus Datura L. International Journal of Applied Research in Natural Products, 1, 15–23.
Sharma, M., Dhaliwal, I., Rana, K., Delta, A. K., & Kaushik, P. (2021). Phytochemistry, pharmacology, and toxicology of Datura species—A review. Antioxidants, 10, 1291.
Shifa, D. M., Leyew, A. Y., & Jufar, M. T. (2024). Datura stramonium seed ingestion leading to unintentional poisoning in a 3‑year‑old Ethiopian toddler: Case report. International Journal of Emergency Medicine.
Steenkamp, P., Harding, N., van Heerden, F., & van Wyk, B.‑E. (2004). Fatal Datura poisoning: Identification of atropine and scopolamine by HPLC/photodiode array/mass spectrometry. Forensic Science International, 145, 31–39.