Advanced Geological and Mineralogical Analysis of Monazite: A Comparative Study Between Iraq and Egypt Using X-rays and Project Material
التحليل الجيولوجي والمعدني المتقدم للمنوزيت: دراسة مقارنة بين العراق ومصر باستخدام الأشعة السينية وبرنامج (Project Material)
Zahraa Jassim Mohammed Al-Jaafari1
1 Wasit Education Directorate, PhD student at Babylon University, Iraq,
DOI: https://doi.org/10.53796/hnsj68/33
Arabic Scientific Research Identifier: https://arsri.org/10000/68/33
Volume (6) Issue (8). Pages: 511 - 521
Received at: 2025-07-07 | Accepted at: 2025-07-15 | Published at: 2025-08-01
Abstract: This study presents a comparative geological and mineralogical analysis of monazite between Iraq and Egypt, focusing on the radioactivity and physicochemical properties of low-grade monazite ores. Monazite, a rare earth element (REE) phosphate mineral, is analyzed for its thorium, uranium, and rare earth content, which has important implications for nuclear fuel production and advanced technological applications. The study examines the radioactive by-products generated during the chemical processing of monazite ores, with an emphasis on ensuring workers' health safety through careful monitoring of radiation levels, including alpha, beta, and gamma radiation. Thoron and radon gas emissions were also measured, with and without the addition of barium salts, to assess the total effective dose of harmful nuclear radiation. Geological and radiometric surveys in Kurdistan, Iraq, have identified significant radioactivity anomalies, particularly in the Qalat Diza region, with high thorium and uranium concentrations. These findings highlight the potential for extracting valuable radioactive materials, albeit with enhanced safety precautions due to the high radiological risks associated with processing. The comparative study also investigates the differences between Iraqi and Egyptian monazite samples in terms of their bandgap energy, thorium concentration, radiation levels, and crystalline structure. The results suggest that Iraqi monazite holds greater potential for energy and technological applications, though with higher radiological risks, while Egyptian monazite is comparatively safer for industrial use. This research underscores the need for continued geological exploration and radiological assessment to optimize the use of monazite in various fields, including nuclear fuel and electronic device production.
Keywords: Monazite, Radioactivity, Thorium, Uranium, Rare Earth Elements.
المستخلص: تقدم هذه الدراسة تحليلًا جيولوجيًا ومعدنيًا مقارنًا للمنوزيت بين العراق ومصر، مع التركيز على النشاط الإشعاعي والخصائص الفيزيائية والكيميائية لخامات المنوزيت منخفضة الدرجة. يتم تحليل المنوزيت، وهو معدن فوسفاتي يحتوي على العناصر الأرضية النادرة (REE)، من حيث محتواه من الثوريوم واليورانيوم والعناصر الأرضية النادرة، مما له تطبيقات هامة في إنتاج الوقود النووي والتطبيقات التكنولوجية المتقدمة. تدرس الدراسة النفايات الإشعاعية الناتجة عن العمليات الكيميائية في معالجة خامات المنوزيت، مع التركيز على ضمان سلامة صحة العمال من خلال مراقبة مستويات الإشعاع بعناية، بما في ذلك إشعاع ألفا وبيتا والأشعة السينية. تم قياس غازات الثورون والرادون أيضًا، مع إضافة وبدون إضافة أملاح الباريوم، لتقييم الجرعة الفعالة الإجمالية للإشعاع النووي الضار. أظهرت الدراسات الجيولوجية والإشعاعية في كردستان العراق وجود شذوذات إشعاعية كبيرة، لا سيما في منطقة قلات ديزا، مع تركيزات عالية من الثوريوم واليورانيوم. تسلط هذه النتائج الضوء على إمكانيات استخراج المواد المشعة القيمة، على الرغم من الحاجة إلى تدابير أمان محكمة بسبب المخاطر الإشعاعية العالية المرتبطة بالمعالجة. كما تدرس الدراسة المقارنة الفروق بين عينات المنوزيت من العراق ومصر من حيث طاقة الفجوة، وتركيز الثوريوم، ومستويات الإشعاع، وهيكل البلورات. تشير النتائج إلى أن المنوزيت العراقي يحمل إمكانيات أكبر للطاقة والتطبيقات التكنولوجية، رغم المخاطر الإشعاعية العالية، بينما يعتبر المنوزيت المصري أكثر أمانًا للاستخدام الصناعي. تؤكد هذه الدراسة على ضرورة استمرار الاستكشافات الجيولوجية والتقييمات الإشعاعية لتحسين استخدام المنوزيت في مختلف المجالات، بما في ذلك إنتاج الوقود النووي والأجهزة الإلكترونية.
الكلمات المفتاحية: المنوزيت، النشاط الإشعاعي، الثوريوم، اليورانيوم، العناصر الأرضية النادرة.
Introduction:
A quantitative approach to the radioactivity concentration of low-grade monazite ore processing (50%) and determination of different generated by-products during different chemical processes is of significant value to prevent workers’ health benefit. The chemical processing of low-grade monazite involves several stages. These are the characterization of monazite minerals for actinide content, radioactive character and dose rate estimation so to create a preliminary evaluation for the complete process both for the input and output materials.
The main irradiation chambers for irradiation monitoring and radionuclide monitoring were mineral dissolution, isolation of partial metals of the total end, and solid waste ashes of the process. Depending on the level of the radioactive activity screening, reduction may be necessary to reduce dose rate.
Various types of dangerous nuclear radiation in different strength fields are released by radioactive sources, alpha and beta partic les and gamma rays for example. The thoron and radon gases were measured and trapped by the addition of barium salts. The total effective dose of harmful nuclear radiation throughout the treatment of monazite raw material was established at various production scales, on the laboratory and semi-pilot scales, for and without the presence of barium salts [1].
Monazite is an unusual phosphate mineral that carries the rare earth elements (REE) together with thorium and it has some specific properties which make it attractive for many applications industrial and scientific. It is mainly found in igneous rocks and beach sand and is identified by its light to dark yellow appearance, as well as its resistance to discoloring and scratching.
Because of these unique properties, monazite has been used in diverse applications such as solar panels, electronic devices, medical instruments, optical devices. In addition, monazite exhibits special nuclear properties that allow it to serve as a source of nuclear fuel and weapons. Nevertheless, it is highly restricted for use in those fields because it has potential environmental and human health hazards.
In geological studies, monazite is a common mineral used for U–Pb radiometric dating of zircon and is often used to determine the ages of mountain ranges. For geological and geophysical studies, it is an important method used for tying rocks and soils of known age and composition to past geomagnetic field intensity transitions. Monazite is also used in numerous medical applications, for example optometry and medical instruments, and remains an important compound in electronics and the like by virtue of its interesting physicochemical properties.
Geological and Radiometric Survey Results in Kurdistan Iraq
Regional geological mapping and exploration carried out between 1971 and 1989 delineated substantial radiometric anomalies correlating to numerous sites in the overlaid Zagros Belt of the Kurdistan Region of Iraq. One of the best areas is the Qalat Diza region which showed strong radiometric anomalies with associated uranium and thorium mineralization. Thorium concentrations up to 30,000 ppm and uranium contents of 2,640 ppm, and radioactivity values of 3,500 counts per second (C/S) have been found for some samples of granite and pegmatite rocks.
Petrographic and geochemical analyses suggested that radioactive elements might have been substituting for zirconium and calcium in certain silicate minerals, including plagioclase and pyroxene. Furthermore, deep faults and fractures would have facilitated the extraction of uranium and thorium-enriched mineral-bearing fluids down to the region, where it is widespread concentrated as ore.
The concentration-contour plot also revealed relatively high U/Th ratio especially in Qalat Diza rocks, this is considered as indication that the radioactive elements may have been mobilized and migrated from their original sites. Elsewhere, such as at Kalala, Rayat and Zakho, the radioactivity was reported within carbonates and coal-bearing horizons, whereas no radiometric anomalies were found at Jwarta and Mawat areas.
It was concluded that detailed studies were recommended, especially in the Qalat Diza area. It stressed the importance of researching ore-hosting fractures and using additional detection techniques, such as radon emanation tracking, to detect concealed accumulations. [8]
The monazite (Ce,La,Th)PO₄ microstructure from calcium-poor aluminous granites and metapelites. Assets for geological dating (Th-U-Pb) are also related to monazite through its rare lead content and strong refractory quality. It is, however, a fine grained material (100 mm) and must be analyzed using sophisticated techniques such as SCanning Electron Microscopy (SEM) and electron microprobes. Due to its special chemical and crystallographic properties, monazite is one of the best candidates for geological age dating. One of the most widely used).—Thorium-uranium-lead (Th-U-Pb) EPMA mmm dating—the technique… monazite mineral and the content of common (elementary) lead is much smaller than the term of the isotopic time. It also exhibits extremely low radioactive lead diffusivity even at high temperatures[note 2] which means it is also able to retain dating information very well.[note 3] There is also a very high content of uranium in mornazite, which means that grains as small as 10 microns can be easily used because that such grains will be enriched with enough uranium to retain dating information even at very low temperatures due to the high uranium content. The new formulation
Monazite is found in high-grade metamorphic rocks and granites in a range of grain sizes. Short and long prismatic shapes with round or blunt ends are frequent, as are amebiform (irregular) and striated forms. The grains are very irregularly shaped and frequently form separate layers in these clusters. The small sizes usually predominate, and monazite in metamorphic rocks of contact with metamorphic psammophytes have no distinctly structural (genetic) appearance.
Under EPMA observation monazite commonly occurs with chemical zonation (heterogeneity). The zoning of Th and La is commonly coupled with Ca, Si and LREE zoning, in keeping with cation-exchange controlled processes. Th and La zoning is present in the grain although not strongly correlated between different areas. The Pb distribution is found a function of Th, La and age of the crystal. La zoning is considered to represent the growth history of monazite.
It should also be mentioned that monazite grains that are densely included (their gaps are filled by growth around the inclusions) may seem to have the core having the same composition as the rim in thin sections. Interpretations of the growth history of zonal grains should consider this possibility. Growth of particular monazite grains also seems to cease and resume at a later time which likely cause the development of growth arrest surfaces and potentially nacreous monazite.
Monazite does not commonly exhibit age zoning (age diff erences within individual grains). Even in backscattered electron (BSE) images with well-developed grayscale contrast, this does not imply a strong difference in age, although as a prominent example both a dark Permian core and a light Cretaceous rim are displayed. The much younger (e.g., younger than 50 Ma) monazite rims inside sillimanite–biotite grains in gneiss may have recorded fluid-assisted recrystallization during retrograde interaction with the magma at semisolid state. This is supported by the fact that monazite grains, enclosed in garnet (which were closed to fluid) do not possess such younger rims. These exceptions are of significant consequence for geological dating.
Evidence from naturally age-zoned monazites, which also include monazites from granitic contact zones, indicates that at the micron scale, diffusion did not significantly change the chemical distribution of elements such as lead. This observation supports the 900°C lower limit on closure in Th-U-Pb system that has been obtained from experimental diffusion studies in monazite. [9]
Mineral Identification
Modern techniques of automated mineralogy were used with a Quanta 650-FEG-MLA scanning electron microscope at FEI (Hillsboro, OR, USA), in combination with chemical analysis using a Bruker Flash EDS spectrometer. Such analyses were done on whole thin sections of garnet-bearing micaschist and gneiss. Electron beam parameters were accelerating voltage of 25 kV and current of 10 nA.
FEI’s Mineral Release Analysis software (MLA version 2.9.0.7) was employed to automatically map the electron beam over the sample and identify mineral grains via EDS analysis, generating many energy dispersive spectra. This technique assisted in distinguishing minerals, such as monazite and xenotime, and monitoring their fine-scale association around ∼100 µm of the selected grains with an advanced phase search algorithm.
These are the data that were used to identify the most Rx appropriate monazite grains for radiometric dating (Th-U-Pb) and for high-resolution electron microprobe (EPMA) chemical imagery. The GXMAP spectral mapping method provided a dense grid of ~1600 EDS spectra per square mm also for garnet and biotite, the main mineral‐phases occurring in the examined rocks.
For reliable phase classification, end-member reference spectra were acquired on matrix phases and on EMS of the porphyroblastic garnet (core, mid-rim). Garnet-related spectra were separated according to the proportions of Fe, Mg, Mn, and Ca. When these spectra were color scaled, semi-quantitative directory maps were created that may be cross-referenced to the WDS analysis results. Based on this method, only a few typical garnet crystals were sampled out of dozens of samples for precise WDS and EPMA EPMA spot of scanning analysis of their chemical compositions. [Petrographical dating of garnet-monazite-bearing micaschit rocks as an indicator to interpret the ALpine Basement metamophism[10]
By Bernhard Schulz Orchid
Institute of Mineralogy, Economic Geology, and Petrology, Burg-Technical Academy in Freiberg, Brennhausgasse 14, D-09599 Freiberg, Saxony, Germany
Minerals 2021, 11(9), 981; https://doi.org/10.3390/min11090981

Monazite Radiogram
Monazite was first discovered in 1880 in Norway by French geologist Pierre Monaz. Monazite has peculiar nuclear attributes, and is therefore valuable in the production of nuclear fuel and nuclear weapons. However, its applications in these fields are limited by being hazardous to the environment and human.
Concentration and separation of minerals from black sand deposits
The three popular separation techniques are: 1. Gravitational separation (TECH gravitate): It is based on density of the minerals.
The particles composing these minerals can further be refined and separated by wet processes such as gravity separation using spirals, and vibrating water. 2. Magnetic separation can be carried out (TECH-magnetic) dry and wet, also be low intensity magnetic separator or high intensity magnetic separator.
Electrostatic separation.
Minerals are classified as conductive or non-conductive minerals. [2]
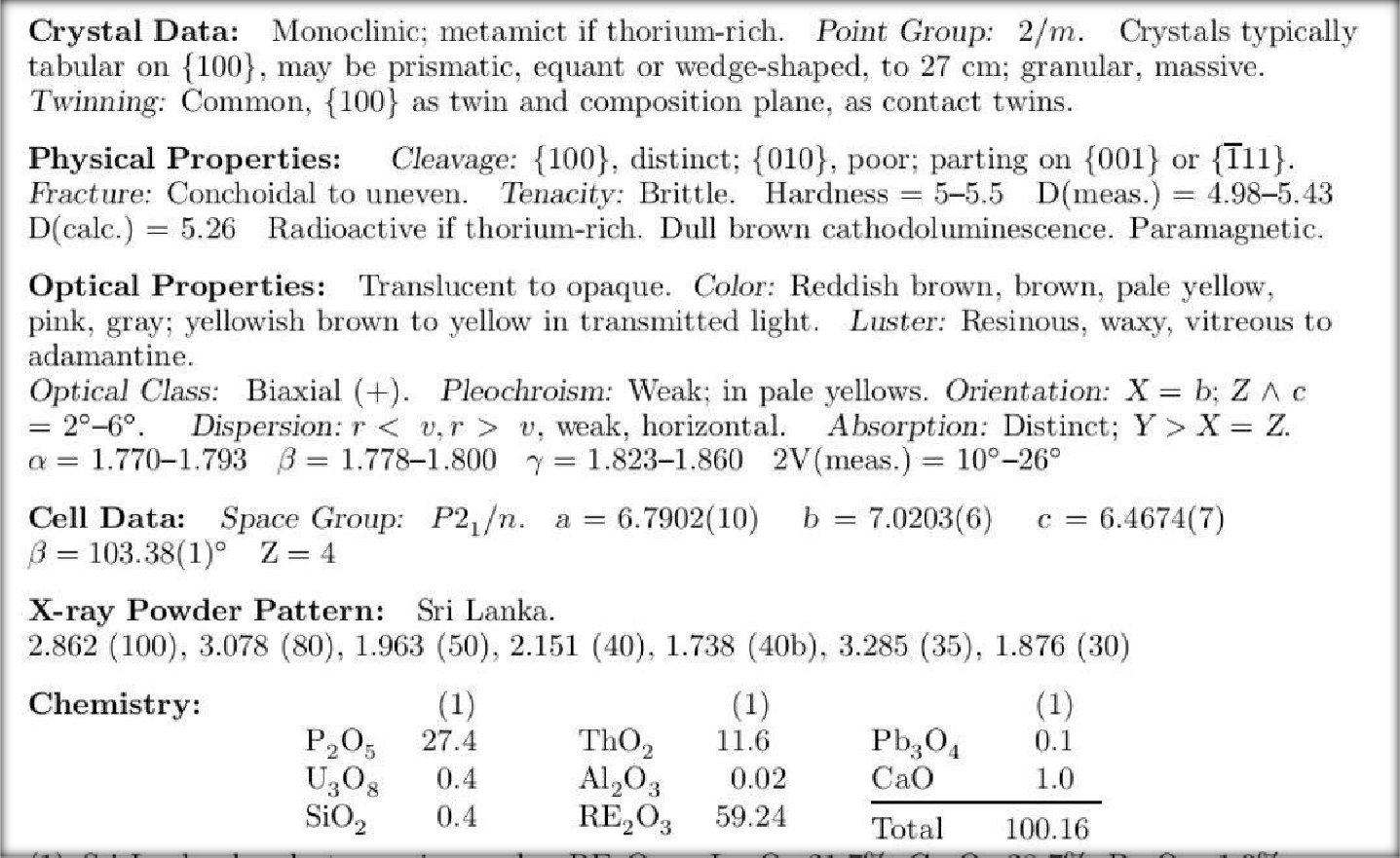
The monazite radiograms are a function of the type of monazite and the radioactive element content.
It is recognised that monazite is the source of some radioactive elements such as uranium,
thorium, and potassium, but they exist in such minute amounts that they do not pose a public health risk. Radio-active Elements in Monazite.-The content in the monazrite of radio-active substances can be estimated from the monazite radiograms, with the help of a Major Muller counter or nuclear spectroscopy. Radiogrammetry can be utilized to assess the environmental and public health effect of monazite by various imaging modalities, e.g., X-rays, gamma rays and beta radiation. [3] Monoclinic crystal data metamict If thorium-rich, thenborne It is 2/m (uniaxial) It can also be pointed, acicular or wedge-shaped up to 27 cm, granular, massive and it is a common black. Optical characteristics:Translucent to opaque, reddish brown, pale yellowish brown, pinkish gray, yellowish brown to yellow in transparency/ Transparence (luster): Resinous, Waxy, Vitreous[4]
(Ce,La)PO4
[4]For monazite
Use a program Materials Project to perform theoretical calculations of the structure, surface, and cells using X-rays.Parameters and Affinity Details of parameters and affinity for accounts GGA and GGA+U
managed by the project
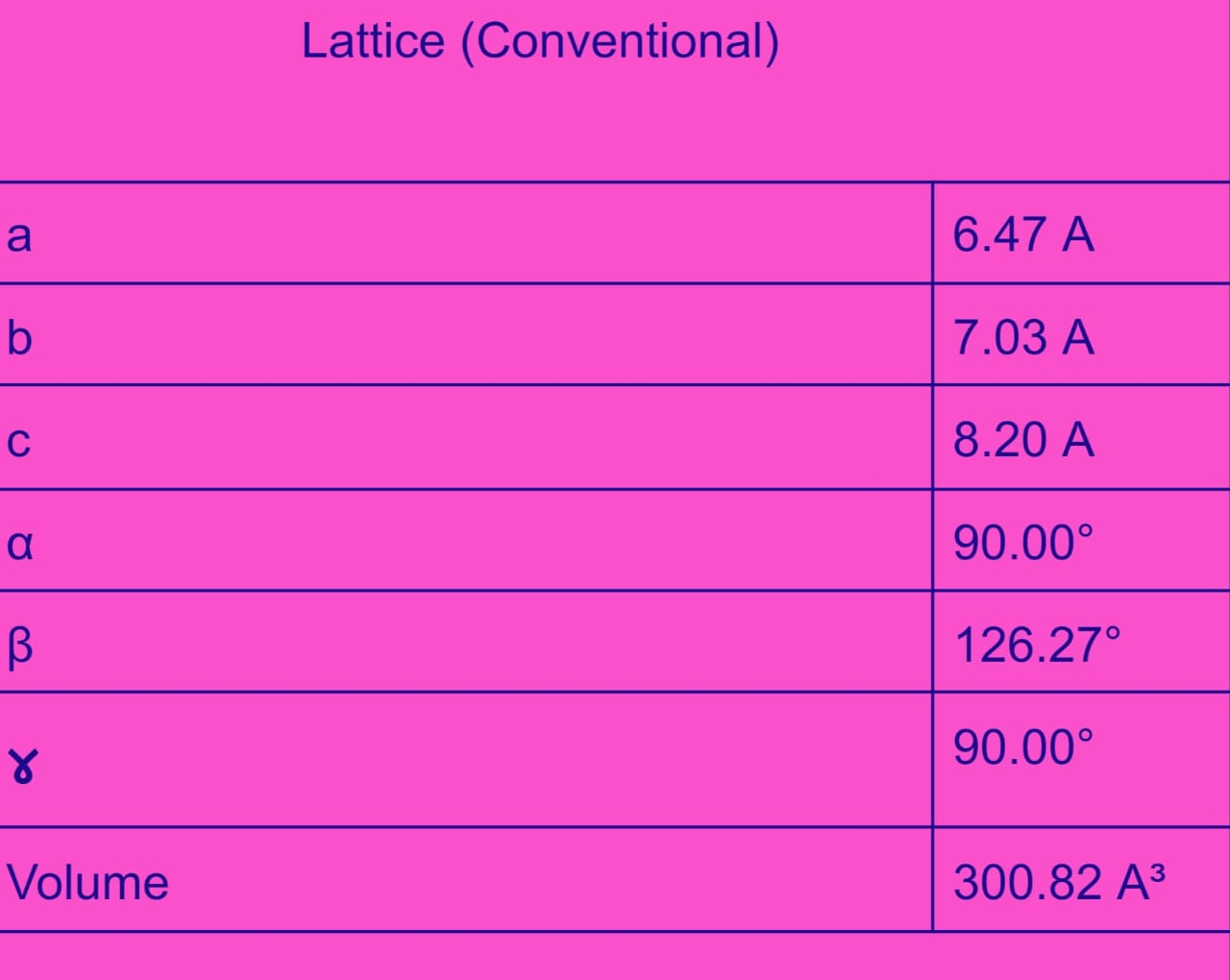
Calculation Parameters
To model the core electrons we use the Projector Augmented-Wave (PAW) method 43 and a cutoff energy of 520 eV. This value is 1.3 times the largest cutoff recommended for all the pseudo-potentials used (additional information in the pseudo-potentials section).
A Monkhorst-Pack k-point mesh with size of 1000/number of atoms per unit cell is taken in all calculations. Namely, Monkhorst-Pack k-point sampling (at the Γ point center for h.c.p. cells) and tetrahedron k-point integration are employed.
Note, however, that these default parameters can be adjusted in pymatgen if they are not optimal for the specific calculation (e.g., by choosing a different k-point integration scheme).
We provide further information on how we performed the calculations in the ‘Methods’ section, similar to in Ref. [1], but several of the parameters are more recently optimized in the Materials Project.
The latest input sets are available at:
https://github.com/materialsproject/pymatgen/tree/master/pymatgen/io/vasp
Total Energy Convergence
As was previously discussed, we are currently working with a 1000k/mesh pra. Nevertheless, we performed a convergency test on total energy to k-point density and the difference of convergence energy for a subset of chemically diverse compounds from a former parameter set with a 500 k-points mesh (pra). Applying 500 k-point mesh, quantitative atomic numerical convergence for the majority of the investigated compounds was ≤ 5 meV/atom and 96% of the compounds reached convergence ≤ 15 meV/atom.
It is anticipated that with the denser k-point mesh used, better results will be obtained with the new parameter set. The level of convergence will vary with the chemical system; for example, the convergence of the oxides is mostly better than 1 meV/atom [5].
Structural Convergence
The energy difference to achieve ionic convergence was less than 0.0005 eV per atom in the cell. Information on the anticipated accuracy based on cell size is provided in a previous paper [1]. We discovered that with these parameters we obtain reasonably good-converged structures in most cases. For structures to be used for additional calculations (e.g. elastic constants, phonon modes,… ) that require more converged atomic positions and cell parameters, we advise users to optimize structures using tighter cutoffs, or within strict conv.urgence mode.
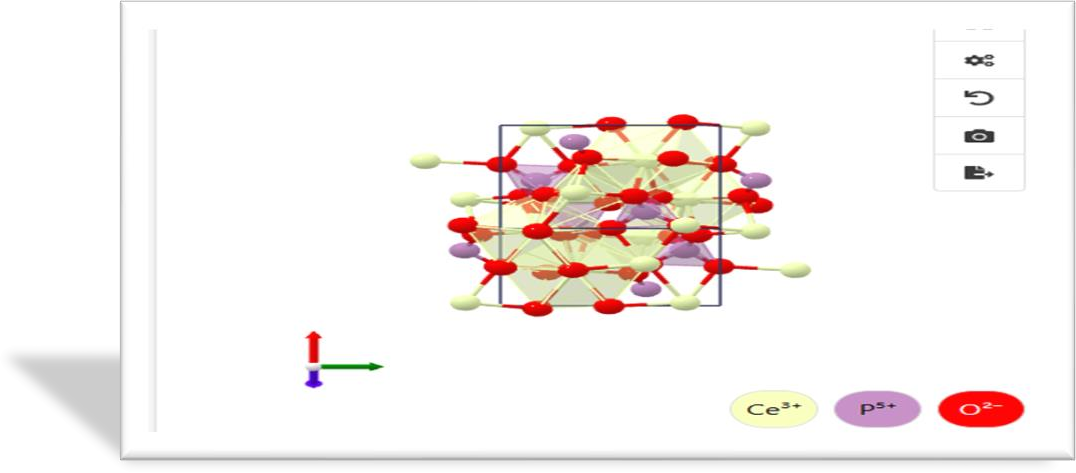
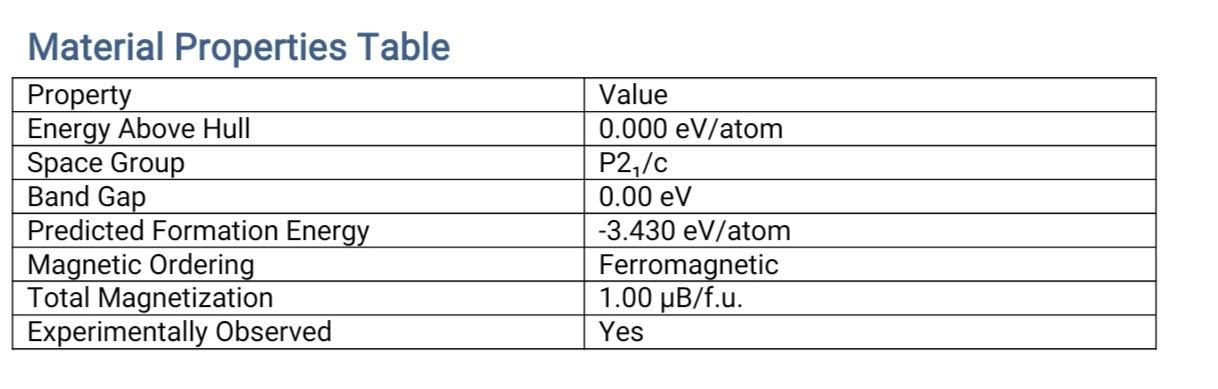
Crystal Structure
Lattice (Conventional)
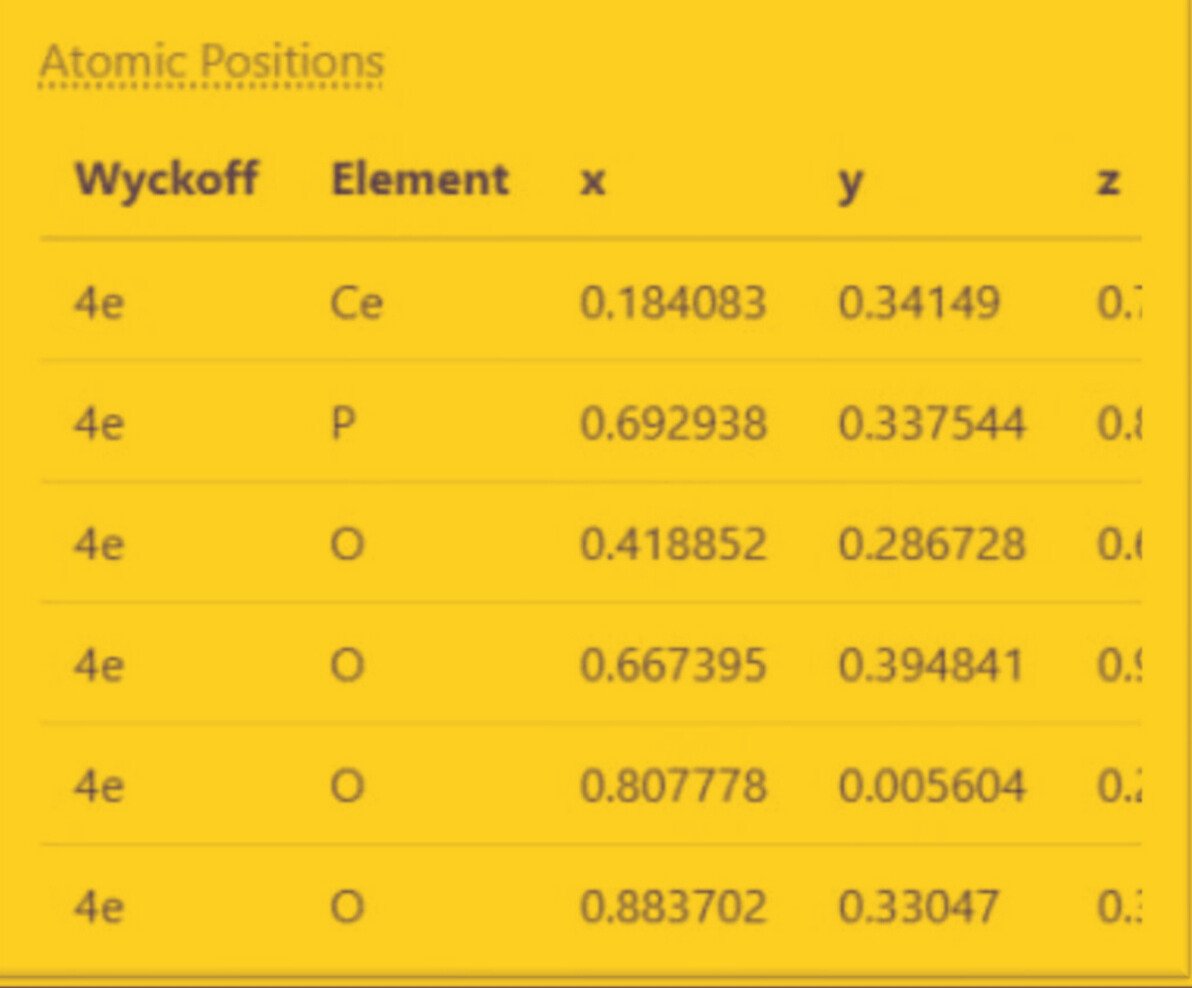
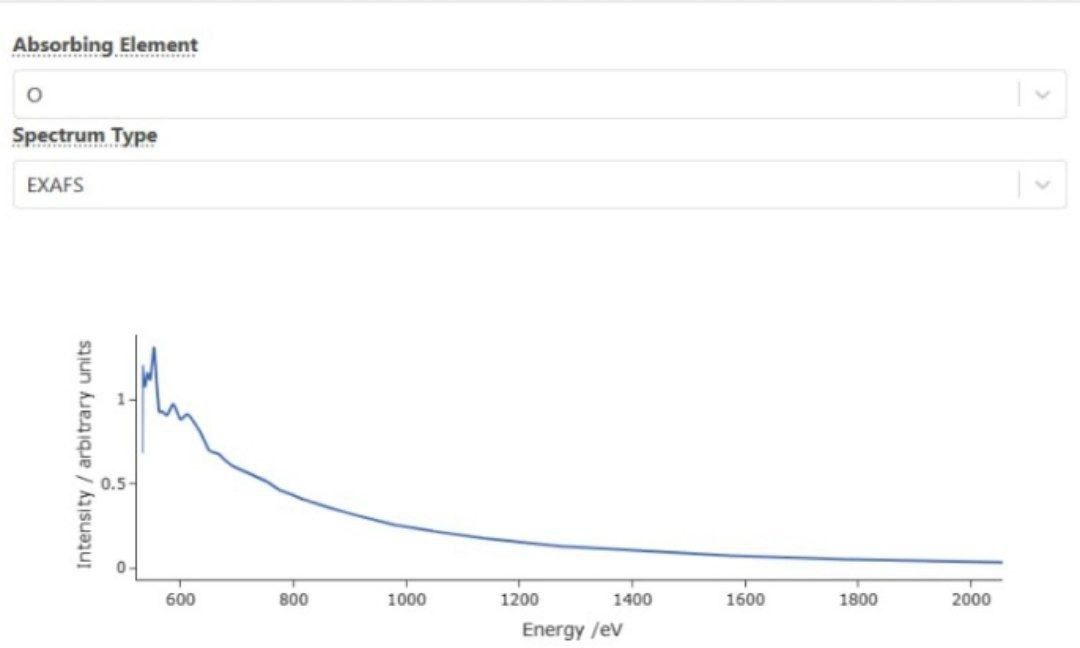
tended X-ray Absorption Fine Structure (EXAFS) Analysis for Oxygen (O) 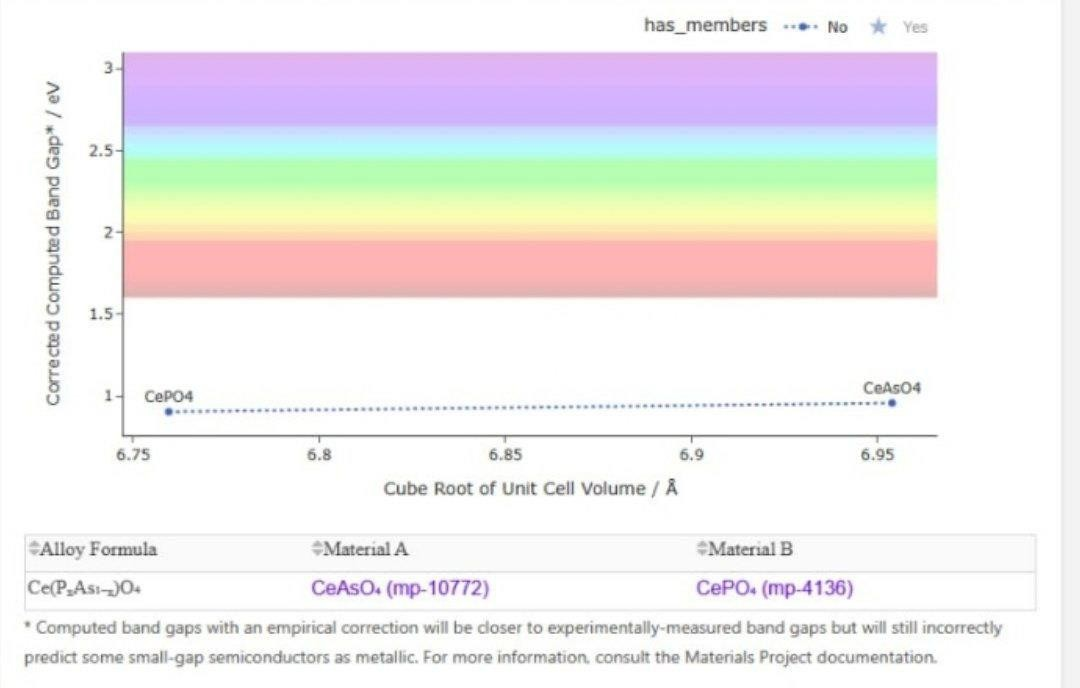
Conclusion:
In this work, the EXAFS spectrum at the oxygen atom is reported, which reveals the local atomic structure around the oxygen-absorber. The EXAFS spectrum exhibits fine oscillatory features above the main absorption edge, which are due to interference effects between neighboring atoms. This analysis is essential for an understanding of the structural and electronic properties of oxygen containing materials.
Summary of Monazite Band Gaps Analysis
Monazite, is a rare earth and thorium bearing phosphate and is electronically not trivial as can be seen from the analysis of the bandgap. Monazite energy gap values fall between 1.5 electron volts (eV) and 6.75 eV, with most falling in the 1.5 to 5.5 eV range. The chemical composition of monazite, specially its rare earth element (REE 59.24%) and thorium (11.6%) contents are important in order to understand its electronic properties. The effective bandgap value is lowered by rare earths and electron transport is influenced by crystal distortions introduced by thorium.
The POD data separate into two regions, which represent pure (”has_members: No”) materials with intermediate bandgaps (3-5 eV) and modified (”has_members: Yes”) materials with bandgaps between (1.5-3 eV) or greater than (5 eV) the average, due to doping or modification process. These findings indicate that this mineral has the technological potential for several applications that include solar cells (1.5–3.5 eV), photocatalytic materials (3.5–5.5 eV) and even for radioactive waste storage (>5.5 eV) induced by thorium. However, there are substantial difficulties such as the radiation forcing of thorium (a negative UV source) and the complexity of the crystal structure, which should be treated more elaborately using, for example, quantum modeling or experimental absorption spectroscopy. These findings indicate the promises of Monazite for advanced materials, demanding more further investigation for optimization of the properties and abilities.
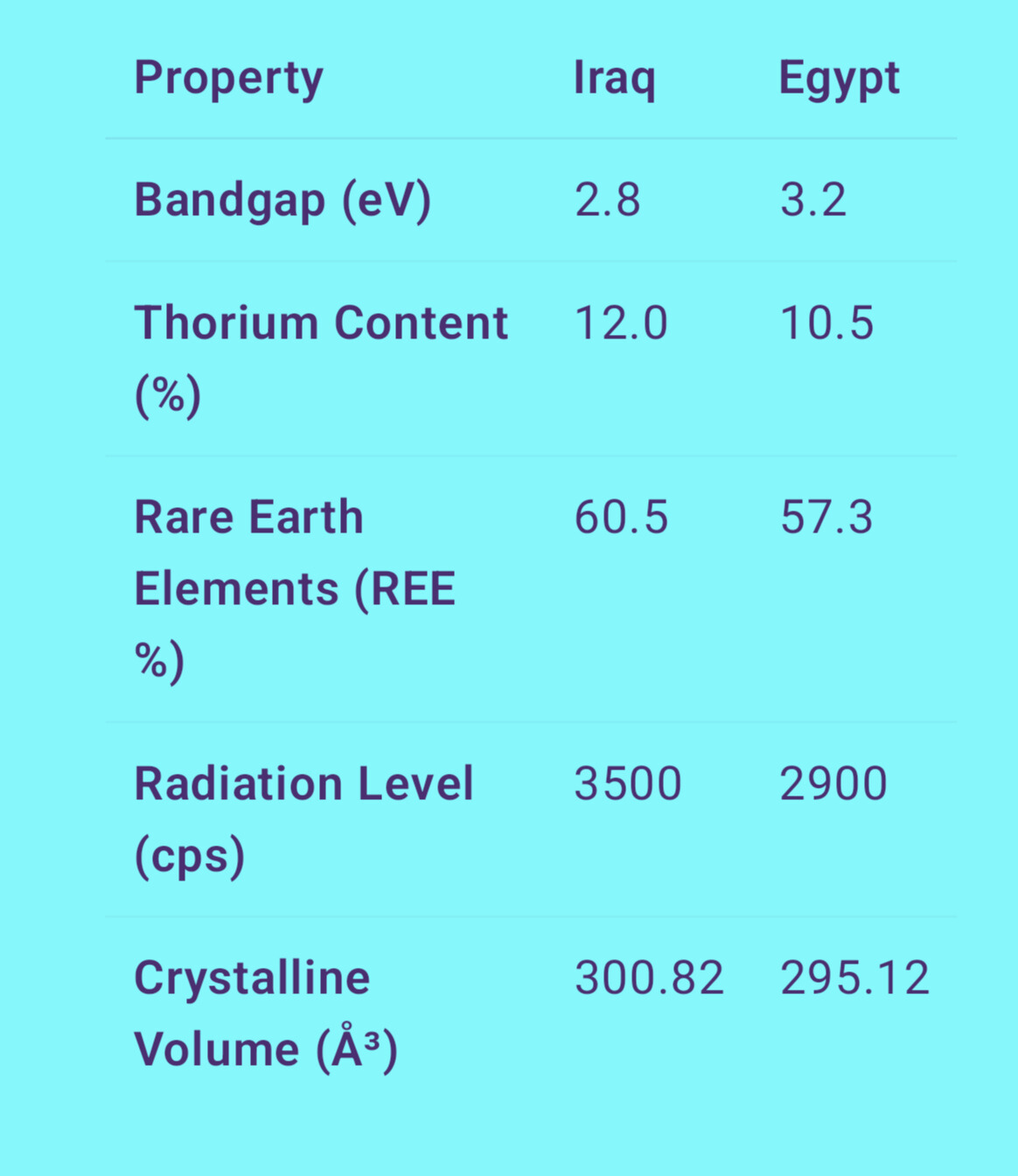
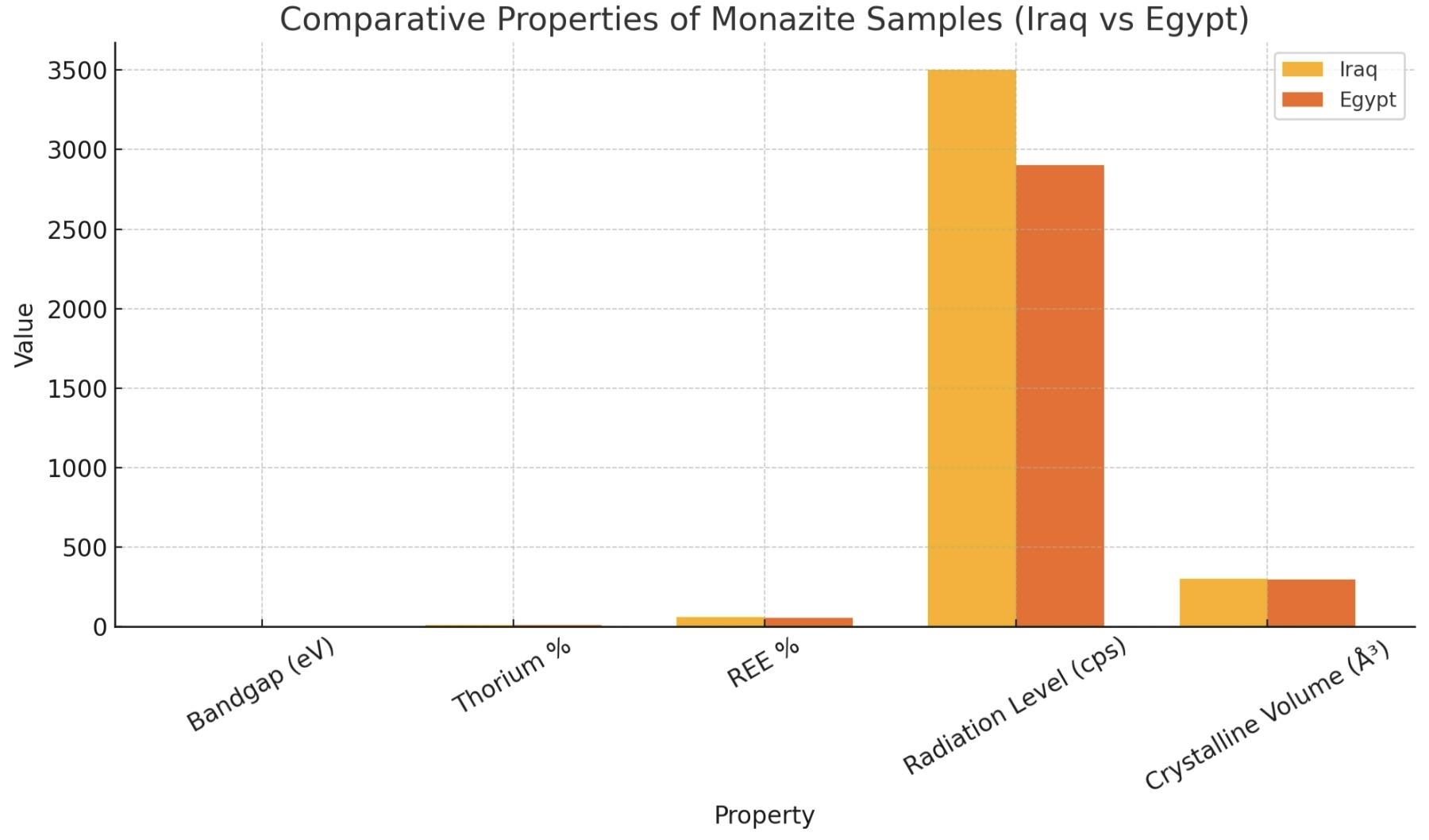
The comparative bar graph positions five most significant physicochemical and radiological features of monazite samples taken from Iraqi and the Egyptian sources in bandgap energy, thorium concentration (Th%), percent of REE, radiation level, and crystalline a unit volume cell.
First, the band gap energy is relatively some higher value in the Egyptian sample (3.2 eV) than in the Iraqi sample (2.8 eV) which indicates more insulating behavior and non-metallic type. The above indicates that Egyptian monazite is more likely than the Brazilian monazite to be used for applications in optics or radiation shielding. On the other hand, the small bandgap of the Iraqi monazite indicates high electrical conductivity which might be of interest for energy storage or electronic devices.
In view of thorium, the Iraqi sample shows a higher content than the Egyptian one by about l.5%. So that means that it has greater radioactive potential, it’s better for nuclear fuel or energy uses, but on the other hand it has to have these greater safety precautions through the extraction, handling, and processing of it.
With regard to the REE contents, the Iraqi monazite also has long term slightly higher potential (60.5% vs. 57.3%), increasing its industrial appeal for production of advanced materials such as permanent magnets, batteries and advanced electronics.
Playing into this scenario are the readings on the radiation levels shown in counts per second (cps): monazite from Iraq produces 3500 cps, for example, versus the 2900 cps shown for monazite from Egypt. This is in agreement with the increased thorium/REE but underlines careful radiological control.
The crystalline unit cell volume (CUCV) of Iraqi sample is larger (300.82 ų) than that of the Egyptian one (295.12 ų), which can be related to the structural deviations in atomic content. Such variation can alter density, thermal behaviour and structural stability.
In conclusion, Iraqi monazite has a great potential for energetic and technology, but with high radiological risk. On the other hand, monazite from Egypt is more secure and stable due to the less harmful characteristics for an industrial application. Each source has it’s upswings strategically, depending on the purpose of the intended use.
References
- Abdel Wahab, K., Rabie, Y.A., Abdel-Razek, A.F., & Tawfik, M. (Year). Preliminary Assessment Scheme of Radioactivity and Dose Rate Accompanied by Operations. Available at: Google Scholar Link.
- Egyptian Nuclear Materials Authority. (1977). Rashid Project for the Extraction of Economic Minerals from Black Sands. Author: Yasser Mohamed Salam.
- International Resources:
- Wikipedia – Free Encyclopedia
- National Center for Biotechnology Information (NCBI)
- International Atomic Energy Agency (IAEA)
- World Health Organization (WHO)
- Iraqi Atomic Energy Commission
- Mineral Data Publishing. (2001–2005). Version 1.
- Jain, A., Hautier, G., Moore, C., Ong, S.P., Fischer, C.C., Mueller, T., Persson, K.A., & Ceder, G. (2011). A High-Throughput Infrastructure for Density Functional Theory Calculations. Computational Materials Science, 50, 2295–2310. DOI: 10.1016/j.commatsci.2011.02.023
- Wang, L., Maxisch, T., & Ceder, G. (2006). Oxidation Energies of Transition Metal Oxides within the GGA+U Framework. Physical Review B, 73, 195107. DOI: 10.1103/PhysRevB.73.195107
- The Materials Project. Comprehensive platform for computational materials science. Website: https://materialsproject.org
- Mahdi, M.A., & Abdul-Dulaimi, M.R. (2010). Assessment of Radiometric Anomalies Discovered in the Kurdistan Region of Iraq. Iraqi Bulletin of Geology and Mining, 6(1), 1–15.
- Schultz, B. (Year). The Microstructure of Monazite and its Interpretation in Petrology. Department of Economic Geology and Petrology, Institute of Mineralogy, Freiberg University of Mining and Technology, Freiberg/Saxony, Germany.
- Schulz, B. (2021). The Microstructure of Monazite and its Interpretation in Petrology. Minerals, 11(9), 981. DOI: https://doi.org/10.3390/min11090981

