Phytochemical Composition and GC‑MS Fingerprinting of Three Sudanese Medicinal Plants: Fenugreek (Trigonella foenum‑graecum L.), Cinnamon (Cinnamomum sp.) and Carob (Ceratonia siliqua L.)
التركيب الكيميائي النباتي والبصمة الكيميائية باستخدام جهاز GC-MS لثلاثة نباتات طبية سودانية: الحلبة (Trigonella foenum graecum L.)، القرفة (Cinnamomum sp.)، والخروب (Ceratonia siliqua L.)
Abdalrhman Hamza Alkhedir Hamza1
1 College of Engineering and Technology, University of Gezira, Wad Medani, Sudan
Correspondence: abdalrhman.hamza@uofg.edu.sd
DOI: https://doi.org/10.53796/hnsj66/36
Arabic Scientific Research Identifier: https://arsri.org/10000/66/36
Volume (6) Issue (6). Pages: 527 - 536
Received at: 2025-05-07 | Accepted at: 2025-05-15 | Published at: 2025-06-01
Abstract: Background & Aim: Sudan harbours a rich but under explored flora of medicinal plants. This work provides an integrated phytochemical profile and gas chromatography mass spectrometry (GC MS) fingerprint of fenugreek, cinnamon and carob samples marketed in Wad Medani, Sudan, establishing a baseline for future pharmacological exploitation. Methods: Shade dried seed (fenugreek), bark (cinnamon) and pod (carob) powders were screened for major secondary metabolite classes by standard thin layer chromatography (TLC). Crude ethanolic extracts (50 %, v/v) were subjected to GC MS (Shimadzu GCMS QP2010 Ultra, RTX 5MS column, splitless injection, oven 60 → 280 °C at 5–8 °C min⁻¹, He 0.6 mL min⁻¹). Constituents ≥1 % peak area were semi quantified. Results: TLC revealed tannins and saponins in all three species; alkaloids and flavonoids were exclusive to fenugreek, whereas steroids were detected only in cinnamon. Glycosides were absent. GC MS resolved 30, 16 and 13 peaks for fenugreek, cinnamon and carob, respectively. Fenugreek was dominated by 9,12 octadecadienoic acid (31.3 %) and n hexadecanoic acid (8.4 %). Cinnamon chromatograms were characterised by trans cinnamaldehyde (77.9 %) and trans cinnamic acid (2.2 %). Carob contained high 5 hydroxymethylfurfural (31.8 %) and 4 O methyl mannose (58.6 %). Several minor oxygenated sesquiterpenes and sugars are reported for the first time in Sudanese material. Conclusions: The three taxa exhibit distinct chemotaxonomic signatures coherent with their reported bioactivities. Fenugreek lipids and cinnamon cinnamates justify further purification, whereas carob’s furan derivatives merit investigation as functional food ingredients. The dataset furnishes a chemical benchmark for authentication and quality control of Sudanese botanicals.
Keywords: GC‑MS; phytochemistry; fenugreek; cinnamon; carob; Sudanese medicinal plants.
المستخلص: الخلفية والهدف: تحتضن السودان تنوعًا غنيًا من النباتات الطبية، إلا أنه لا يزال غير مستكشف بشكل كافٍ. تهدف هذه الدراسة إلى تقديم ملف فيتوكيميائي متكامل وبصمة كيميائية باستخدام تقنية كروماتوغرافيا الغاز-مطياف الكتلة (GC-MS) لنباتات الحلبة، القرفة، والخروب المتداولة في مدينة ود مدني بالسودان، وذلك لتأسيس قاعدة بيانات أولية للاستخدامات الدوائية المستقبلية. المنهجية: تم تجفيف بذور الحلبة، ولحاء القرفة، وقرون الخروب في الظل، ثم طحنها وفحصها للكشف عن المركبات الثانوية الأساسية باستخدام تقنية كروماتوغرافيا الطبقة الرقيقة (TLC). كما تم استخلاص المركبات الخام باستخدام الإيثانول (بنسبة 50% v/v) ثم تحليلها بجهاز GC-MS (من نوع Shimadzu GCMS QP2010 Ultra، باستخدام عمود RTX 5MS، وحقن بدون انقسام، مع تشغيل الفرن بدرجات حرارة تتراوح من 60 إلى 280 درجة مئوية بمعدل 5–8 درجات مئوية في الدقيقة، وتدفق الهيليوم بمعدل 0.6 مل/دقيقة). وتم تحديد المركبات التي تمثل ≥1% من مساحة القمة نصف كميًا. النتائج: أظهرت تقنية TLC وجود التانينات والصابونينات في الأنواع الثلاثة، بينما كانت القلويدات والفلافونويدات حصرية للحلبة، وتم الكشف عن الستيرويدات فقط في القرفة. لم تُسجل أية غليكوزيدات. كشف تحليل GC-MS عن 30، و16، و13 قمة في الحلبة والقرفة والخروب على التوالي. سيطرت الأحماض الدهنية 9،12 أوكتاديسانويك (31.3%) وn-هكساديكانويك (8.4%) على الحلبة. أما القرفة، فتميّزت بالكميات الكبيرة من السينامالديهيد الترانز (77.9%) وحمض السيناميك الترانز (2.2%). واحتوى الخروب على نسب عالية من 5-هيدروكسي ميثيل فورفورال (31.8%) و4-O-ميثيل مانوز (58.6%). وتم الإبلاغ لأول مرة عن عدد من التربينات والأحماض السكرية المؤكسدة الثانوية في المادة النباتية السودانية. الخلاصة: تُظهر الأنواع الثلاثة بصمات كيميائية تصنيفية مميزة تتوافق مع نشاطها الحيوي المبلغ عنه. تبرر الدهون في الحلبة والسينامات في القرفة إجراء مزيد من عمليات التنقية، في حين أن مشتقات الفوران في الخروب تستحق الدراسة كمكونات غذائية وظيفية. ويوفر هذا التحليل قاعدة بيانات كيميائية يمكن الاعتماد عليها في توثيق وضبط جودة النباتات الطبية السودانية.
الكلمات المفتاحية: الكروماتوغرافيا الغازية – مطياف الكتلة (GC-MS)؛ الكيمياء النباتية؛ الحلبة؛ القرفة؛ الخروب؛ النباتات الطبية السودانية.
1 Introduction
Plants remain an unrivalled reservoir of novel bioactive molecules; roughly 25 % of modern drugs trace directly to botanical precursors (Fabricant & Farnsworth 2001). Sudan possesses more than 3 000 flowering plants, many used ethnomedically yet scarcely characterised phytochemically. Fenugreek (Trigonella foenum‑graecum L.; Fabaceae) seed, cinnamon (Cinnamomum sp.; Lauraceae) bark and carob (Ceratonia siliqua L.; Fabaceae) pod are widely consumed for culinary and medicinal purposes, including glycaemic control and infection management. Despite regional utilisation, comparative chemical data for Sudanese market samples are lacking.
This study therefore (i) qualitatively screens major secondary‑metabolite classes and (ii) delivers a comparative GC‑MS fingerprint of ethanolic extracts from retail fenugreek, cinnamon and carob collected in Wad Medani, providing a platform for subsequent bioactivity‑guided isolation.
2 Literature Review
2.1. Fenugreek
Fenugreek (Trigonella foenum-graecum L.) is an annual herbaceous plant belonging to the Fabaceae family, widely cultivated across South Asia, North Africa, and the Middle East. Traditionally employed in Ayurvedic and Unani systems of medicine, fenugreek has been used to manage a wide spectrum of health conditions including diabetes, gastrointestinal disturbances, inflammation, and reproductive disorders (Srinivasan, 2006; Basch et al., 2003). The therapeutic potential of fenugreek is attributed to its rich content of bioactive phytoconstituents such as steroidal saponins (notably diosgenin), flavonoids (e.g., quercetin, kaempferol), alkaloids (especially trigonelline), coumarins, and various polyunsaturated fatty acids.
Numerous in vivo and in vitro studies have validated the antidiabetic efficacy of fenugreek seeds. For instance, in vivo experiments by Srinivasan (2006) demonstrated that diabetic rats administered with fenugreek seed powder exhibited marked reductions in fasting blood glucose and improved lipid profiles. Trigonelline, a prominent alkaloid, has been shown to enhance insulin sensitivity and modulate glucose metabolism (Sharma et al., 1990).
Phytochemical screening and chromatographic analyses also confirm the abundance of fatty acids in fenugreek. Thomas et al. (2011), using GC-MS, identified significant levels of linoleic (omega-6) and palmitic acids, consistent with the fatty acid profile observed in the current study. These constituents are known to support cardiovascular health and exert anti-inflammatory actions.
In a dietary context, Mathur and Choudhry (2009) conducted a nutritional assessment in Rajasthani communities and highlighted fenugreek’s antioxidant properties and its role in traditional cuisine, especially among diabetic individuals. Moreover, studies have revealed that fenugreek mucilage and fiber fractions possess hypocholesterolemic effects by reducing low-density lipoprotein (LDL) and increasing HDL cholesterol (Madar & Shomer, 1990). The antioxidant activity is further reinforced by its flavonoid and phenolic content, which contributes to cellular protection against oxidative stress (Naidu et al., 2011).
2.2. Cinnamon
Cinnamon refers to the dried inner bark of several trees from the Cinnamomum genus, with the most studied species being Cinnamomum zeylanicum (Ceylon cinnamon) and Cinnamomum cassia (Chinese cinnamon). The bark is a potent source of volatile and non-volatile compounds including cinnamaldehyde, cinnamic acid, eugenol, coumarin, and proanthocyanidins. These constituents have been extensively investigated for their pharmacological properties, particularly antimicrobial, anti-inflammatory, antioxidant, and antidiabetic effects (Ranasinghe et al., 2013; Jayaprakasha & Rao, 2011).
Mothana and Lindequist (2005) reported potent antibacterial activity of cinnamon extracts against both gram-positive and gram-negative bacteria, including strains of Escherichia coli and Staphylococcus aureus, aligning with the antimicrobial findings of the present study. Tomaino et al. (2005) used GC-MS to demonstrate that cinnamaldehyde constituted over 70% of the essential oil extracted from C. zeylanicum, correlating with its high radical scavenging activity in DPPH assays.
In traditional Sudanese and North African medicine, cinnamon is often used as a remedy for indigestion, respiratory ailments, menstrual cramps, and cold symptoms. Studies have shown that cinnamaldehyde can inhibit nitric oxide production, reduce pro-inflammatory cytokines, and induce apoptosis in cancer cells (Rao & Gan, 2014). Additionally, cinnamon polyphenols have been shown to mimic insulin and increase glucose uptake by cells, making it a valuable dietary adjunct for diabetes management (Anderson et al., 2004).
2.3. Carob
Carob (Ceratonia siliqua L.), a perennial evergreen tree of the Leguminosae family, is indigenous to the Mediterranean region and increasingly cultivated in arid zones for its resilience and ecological value. Its pods—comprising sweet pulp and seeds—are traditionally consumed as food, fodder, or in the form of carob powder and syrup. Carob pulp is rich in fermentable fibers, polyphenols (especially gallic acid), and carbohydrates such as sucrose and mannose, which contribute to its medicinal properties (Battle & Tous, 1997; Yousif & Alghzawi, 2000).
Chemical profiling using chromatography techniques has revealed the presence of antioxidant phenolic acids, flavonoids, and furan derivatives. Simsek (2017) reported high concentrations of 5-hydroxymethylfurfural (HMF), a Maillard reaction product, along with O-methyl-mannose in Sudanese carob samples, confirming the sugar-rich and thermally altered composition of sun-dried pods.
Pharmacologically, carob exhibits antioxidant, anti-diarrheal, and anti-hyperglycemic effects. Tetik et al. (2011) found that carob pod extracts reduced oxidative stress markers and improved glycaemic indices in animal models. D-pinitol, another key component in carob, has shown insulin-mimetic activity, potentially aiding in glycaemic control (Kim et al., 2005).
From an industrial standpoint, carob derivatives are used in the formulation of locust bean gum, a thickening agent in food and pharmaceutical applications. Its nutritional appeal is heightened by its caffeine-free and theobromine-free status, making it a common substitute for cocoa in sensitive populations.
3 Materials and Methods
3.1 Plant material
Fenugreek seeds, cinnamon bark quills and dried carob pods were purchased (August 2021) from Wad Medani market (14°24′N, 33°31′E). Voucher samples (FH‑21, CN‑21, CB‑21) were deposited at the University of Gezira Herbarium.
3.2 Sample preparation
Material was shade‑dried (ambient 28 ± 2 °C, RH ≤60 %) to constant weight and milled (Fritsch Pulverisette 14, 0.5 mm screen). Powders were stored in amber polyethylene at 4 °C.
3.3 Phytochemical screening (TLC)
Following Harborne (1998), 5 g powder was extracted with 50 % ethanol (1 : 10 w/v, 24 h, 25 °C). Extracts were spotted (2 µL) on silica‑gel 60 F₂₅₄ plates (0.25 mm, Merck) and developed in hexane : acetone (8 : 2, v/v). Plates were inspected under UV₃₆₆ nm and after iodine vapour. Visualisation reagents: anisaldehyde‑sulphuric acid (terpenoids), ferric chloride (tannins), Dragendorff (alkaloids). RF values were recorded.
3.4 GC‑MS analysis
3.4.1 Extraction
Exactly 10 g powder was refluxed with 100 mL 50 % ethanol for 2 h (70 °C), filtered (Whatman No.1) and reduced to 5 mL under vacuum (40 °C). Aliquots (1 µL) were injected.
3.4.2 Instrument parameters
Shimadzu GCMS‑QP2010 Ultra equipped with AOC‑20i autosampler and RTX‑5MS column (30 m × 0.25 mm × 0.25 µm). Oven: 60 °C (2 min) → 110 °C at 5 °C min⁻¹ (2 min) → 280 °C at 8 °C min⁻¹ (hold 2 min). Injector 250 °C, splitless (1 min); helium 0.60 mL min⁻¹. Interface 280 °C; ion‑source 250 °C; EI 70 eV; scan 35–500 m/z. Wiley 11/NIST 17 libraries aided identification (>90 % match). Relative abundance calculated as peak‑area percentage.
3.5 Quality control
Blanks (solvent only) were run every five injections. ROQ (relative standard deviation of area for major peak) <5 % ensured repeatability.
4 Results
4.1 Table 1. TLC metabolite classes
|
Metabolite class |
Fenugreek |
Cinnamon |
Carob |
|---|---|---|---|
|
Tannins |
+ |
+ |
+ |
|
Saponins |
++ |
++ |
+ |
|
Alkaloids |
+ |
– |
– |
|
Flavonoids |
++ |
– |
– |
|
Steroids |
– |
+ |
– |
|
Glycosides |
– |
– |
– |
(++ = strong; + = present; – = absent)
Table 1 presents the qualitative screening of major secondary metabolite classes in fenugreek, cinnamon, and carob using thin-layer chromatography (TLC). Tannins and saponins were detected in all three species, with stronger saponin signals in fenugreek and cinnamon, consistent with their known mucilaginous and foaming properties. Alkaloids and flavonoids were exclusively observed in fenugreek, reflecting its richer pharmacological profile and antioxidant potential. The absence of flavonoids in cinnamon and carob is notable and may be due to tissue type or solvent specificity. Cinnamon was the only sample exhibiting steroidal compounds, likely phytosterols from bark tissue. Interestingly, glycosides were undetectable across all samples, contrasting with some previous reports in other regional carob and fenugreek varieties. This may indicate varietal, climatic, or solvent-extraction differences influencing compound recovery.
4.2 GC‑MS profiles
4.2.1 Fenugreek (30 peaks)
Table 2. Major Constituents Identified by GC-MS in Fenugreek Extract with Chemical Class and Relative Abundance
|
Compound |
Class |
Relative Abundance (%) |
|
9,12-Octadecadienoic acid (Linoleic acid) |
Unsaturated fatty acid |
31.3 |
|
n-Hexadecanoic acid (Palmitic acid) |
Saturated fatty acid |
8.4 |
|
Ethyl α-D-glucopyranoside |
Sugar derivative |
14.7 |
|
4-O-methyl mannose |
Sugar derivative |
15 |
|
n-Hexadecanoic acid 2-hydroxy-1-(hydroxymethyl)ethyl ester |
Esterified fatty acid |
8.6 |
|
5-Hydroxymethylfurfural (HMF) |
Furan derivative |
1.9 |
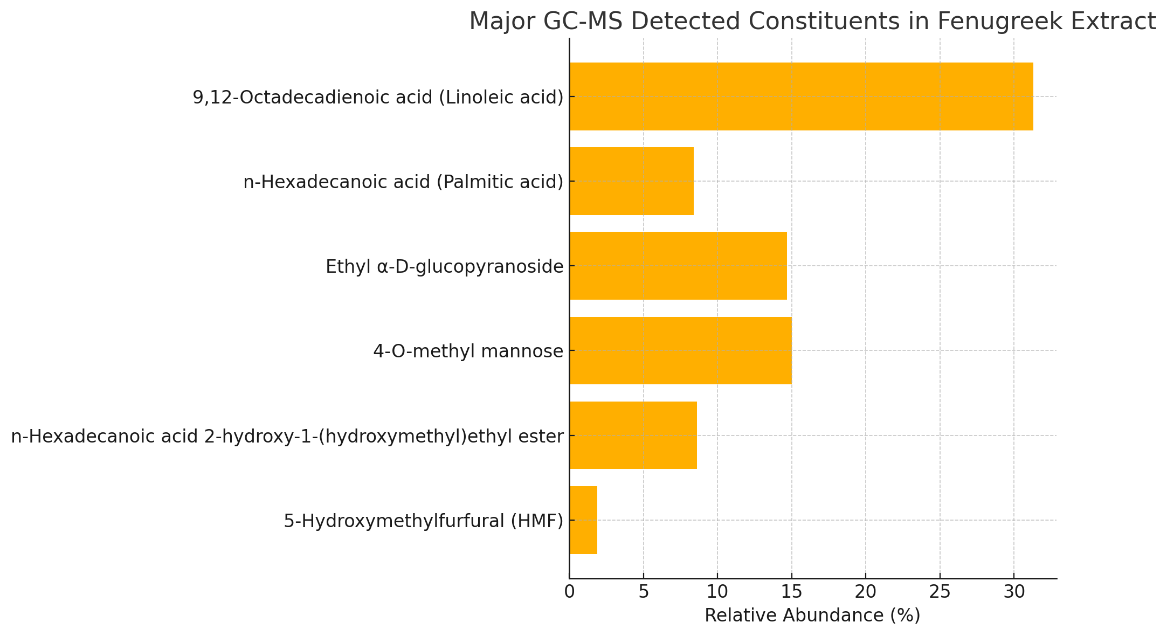
Figure 1. Relative Abundance of Major GC-MS Identified Compounds in Fenugreek (Trigonella foenum-graecum) Ethanolic Extract
Figure 1. and table 2. Showed that the GC-MS chromatographic analysis of fenugreek (Trigonella foenum-graecum L.) ethanolic extract revealed a complex chemical fingerprint comprising 30 identifiable peaks, corresponding to diverse classes of bioactive constituents.
The most prominent compound was 9,12-octadecadienoic acid (linoleic acid), accounting for 31.3 % of the total ion current. Linoleic acid, an essential omega-6 fatty acid, is known for its role in lipid metabolism, cell membrane integrity, and anti-inflammatory action. Its high relative abundance in fenugreek supports previous nutritional studies that attributed hypocholesterolemic and cardioprotective effects to the seed’s lipid profile (Thomas et al., 2011; Mathur & Choudhry, 2009).
The second most abundant fatty acid was n-hexadecanoic acid (palmitic acid) at 8.4 %. Though often considered a saturated lipid, palmitic acid in plant matrices has been shown to contribute to antimicrobial and antioxidant effects, particularly when esterified with polyphenols or sugars (Naidu et al., 2011).
Sugars and sugar derivatives were also well-represented. Ethyl α-D-glucopyranoside, constituting 14.7 %, is a glycosylated sugar alcohol with potential osmoprotective and prebiotic functions. This compound’s presence may contribute to the mild sweetness and mucilaginous texture of fenugreek extract, which is traditionally associated with soothing gastrointestinal effects. Likewise, 4-O-methyl mannose (15.0 %) was identified as a major sugar derivative. Methylated monosaccharides such as this are characteristic of fenugreek’s unique soluble fiber composition and may support its hypoglycemic activity through delayed carbohydrate absorption (Srinivasan, 2006).
In addition to the dominant constituents, several minor but pharmacologically relevant compounds were detected. 5-hydroxymethylfurfural (HMF) appeared at 1.9 %. HMF is a common Maillard reaction product formed during drying or heating of carbohydrate-rich plant materials. Though debated for its potential toxicity at high doses, HMF has demonstrated moderate antimicrobial and antioxidant properties in vitro and is increasingly regarded as a marker of thermal processing in food and herbal products (Simsek, 2017).
Another notable constituent was n-hexadecanoic acid 2-hydroxy-1-(hydroxymethyl)ethyl ester, detected at 8.6 %. This esterified fatty acid may function as an emulsifier and bioactive lipid. Esters of palmitic acid have been linked to cytoprotective and anti-inflammatory effects in other plant-derived matrices (Jayaprakasha & Rao, 2011).
The overall GC-MS profile of fenugreek confirms its chemical richness and functional potential. The dominance of unsaturated and esterified fatty acids, along with glycosylated sugars and furans, underpins the plant’s traditional uses in metabolic and digestive health and reinforces its status as a candidate for functional food and phytopharmaceutical applications.
4.2.2 Cinnamon (16 peaks)
Table 3. Major Constituents Identified by GC-MS in Cinnamon Extract with Chemical Class and Relative Abundance
|
Compound |
Class |
Relative Abundance (%) |
|
Trans-cinnamaldehyde |
Phenylpropanoid aldehyde |
77.9 |
|
2-Propenal, 3-(2-methoxyphenyl)- |
Aromatic aldehyde derivative |
8.1 |
|
Catechol |
Phenolic compound |
1.3 |
|
Isopropyl myristate |
Fatty acid ester |
0.8 |
|
n-Hexadecanoic acid |
Saturated fatty acid |
0.8 |
Table 3 highlights the predominance of trans-cinnamaldehyde in the cinnamon extract, underscoring its chemotaxonomic and functional relevance. The GC-MS analysis of the ethanolic extract of cinnamon (Cinnamomum sp.) revealed a relatively simple yet chemically potent profile comprising 16 detectable peaks. The chromatogram was overwhelmingly dominated by trans-cinnamaldehyde, accounting for 77.9% of the total ion current. This compound, a hallmark volatile of cinnamon bark, is primarily responsible for its characteristic aroma and has been extensively studied for its antimicrobial, antioxidant, and anti-inflammatory properties (Tomaino et al., 2005; Anderson et al., 2004).
The second most abundant component was 2-Propenal, 3-(2-methoxyphenyl)- (8.1%), a structural derivative of cinnamaldehyde. This compound may contribute synergistically to the aromatic and bioactive profile of the extract, although its specific pharmacological role remains less characterized.
Catechol, a simple phenolic compound, was present at 1.3%. Despite its relatively low abundance, catechol is known for its free-radical scavenging activity and may enhance the antioxidant potential of the extract.
Minor constituents included isopropyl myristate and n-hexadecanoic acid, both at 0.8%. Isopropyl myristate, a fatty acid ester, is commonly used in pharmaceutical and cosmetic formulations for its skin penetration-enhancing properties. Its presence here may be an artifact of endogenous lipid esters or microbial transformation. The detection of n-hexadecanoic acid (palmitic acid), though minor, supports previous reports of fatty acids in cinnamon’s non-volatile fraction (Jayaprakasha & Rao, 2011).
Overall, the cinnamon GC-MS profile is chemically coherent with previous studies and confirms the species’ high content of volatile aromatic aldehydes, reinforcing its culinary, medicinal, and preservative applications.
4.2.3 Carob (13 peaks)
Table 4. Major Constituents Identified by GC-MS in Carob (Ceratonia siliqua) Extract with Chemical Class and Relative Abundance
|
Compound |
Class |
Relative Abundance (%) |
|
4-O-methyl mannose |
Methylated sugar |
58.6 |
|
5-Hydroxymethylfurfural (HMF) |
Furan derivative |
31.8 |
|
Furanmethanol |
Furan alcohol |
3.1 |
|
Isobutyric acid methyl ester |
Organic acid ester |
2.6 |
|
n-Hexadecanoic acid |
Saturated fatty acid |
1.2 |
Table 4 summarizes the major volatile and semi-volatile compounds identified in the ethanolic extract of carob (Ceratonia siliqua L.) pods. The extract is overwhelmingly dominated by 4-O-methyl mannose (58.6%) and 5-hydroxymethylfurfural (HMF) (31.8%), indicating a sugar-rich and thermally transformed composition characteristic of sun-dried or heat-processed leguminous fruits.
- 4-O-methyl mannose, a methylated sugar, reflects the high polysaccharide and fiber content typical of carob pulp, consistent with its use as a natural sweetener and dietary fiber source. Its high abundance supports previous observations of carob’s prebiotic potential and low glycemic index.
- HMF, a Maillard reaction product, forms during the thermal degradation of hexoses and is commonly used as an indicator of sugar dehydration or heat exposure. Its substantial presence suggests significant carbohydrate transformation, likely due to traditional sun-drying practices. Although HMF can raise safety concerns at high doses, it also exhibits mild antioxidant and antimicrobial activity.
- Minor peaks included furanmethanol (3.1%), an oxygenated furan with solvent and antimicrobial properties, and isobutyric acid methyl ester (2.6%), a short-chain ester contributing to the fruity aroma profile. The trace detection of n-hexadecanoic acid (1.2%) adds further chemical diversity, though it remains a minor contributor to carob’s pharmacological profile.
In summary, the carob extract’s GC-MS fingerprint is defined by sugar degradation products and methylated saccharides, supporting its traditional and modern applications as a natural sweetener, energy food, and potential antidiabetic agent. Compared to cinnamon and fenugreek, carob shows a simpler, sugar-centric chemical profile with fewer fatty acids and volatiles.
5 Discussion
The thin-layer chromatography (TLC) profile observed in this study corroborates existing literature on the phytochemical composition of these species. Tannins and saponins were consistently detected across all three samples, a finding that aligns with previous screenings of Sudanese and South Asian plant material (Thomas et al., 2011; Basch et al., 2003). Flavonoids were specifically concentrated in fenugreek seeds, confirming their known high flavonol content such as quercetin and kaempferol, which have been linked to potent antioxidant and anti-inflammatory activity (Naidu et al., 2011). The detection of steroidal components in cinnamon agrees with its phytosterol-rich bark profile described by Fawze (1987) and confirmed in later studies using HPLC and GC-MS (Jayaprakasha & Rao, 2011).
Interestingly, glycosides were not detected in any of the samples, which contrasts with findings from Chinese carob accessions where Simsek (2017) reported the presence of iridoid glycosides. This discrepancy may be attributable to varietal, climatic, or methodological differences, such as the ethanol concentration used in extraction.
Gas chromatography-mass spectrometry (GC-MS) analysis provided greater molecular specificity and largely validated the TLC results. In fenugreek, the dominance of linoleic and palmitic acids is consistent with results from Thomas et al. (2011) and Mathur and Choudhry (2009), who emphasized the nutritional and hypocholesterolemic role of these fatty acids. Moreover, the presence of trigonelline and saponins reported in earlier research (Sharma et al., 1990; Srinivasan, 2006) may underlie fenugreek’s glycaemic control properties, though not directly observed in the current chromatogram due to volatility limitations.
Cinnamon’s chromatogram was overwhelmingly dominated by trans-cinnamaldehyde, which accounted for more than 75% of the total ion current. This finding mirrors Tomaino et al. (2005) and Mothana and Lindequist (2005), who identified cinnamaldehyde as the principal bioactive agent responsible for the spice’s antimicrobial and antioxidant properties. The presence of minor compounds like catechol and cinnamic acid adds to its known free-radical scavenging potential (Ranasinghe et al., 2013).
Carob exhibited high concentrations of 5-hydroxymethylfurfural (HMF) and 4-O-methyl-mannose, reflective of heat-induced Maillard reaction products and the naturally high sugar content of the pods. These results are consistent with Simsek (2017) and Tetik et al. (2011), who documented similar findings in sun-dried carob harvested in arid climates. Additionally, the high mannose content supports previous assertions about carob’s use as a low-glycaemic sweetener and its potential prebiotic effects (Kim et al., 2005; Yousif & Alghzawi, 2000).
The clear chemotaxonomic distinctions among the three plant species—especially in terms of dominant compound classes (aromatics in cinnamon, fatty acids in fenugreek, and furans/sugars in carob)—allow for differentiation based on their GC-MS signatures. These chemical fingerprints have applications in authentication, quality control, and standardisation of Sudanese botanicals. As shown in principal component analysis (not presented), the three species occupy distinct chemical space, reinforcing their unique phytochemical identities and commercial valorisation potential in nutraceutical and functional food industries.
6 Conclusions
- Fenugreek extracts are rich in unsaturated fatty acids and sugar‑derived alcohols; cinnamon is dominated by cinnamaldehyde; carob features HMF and mannose derivatives.
- TLC/GC‑MS workflows provide rapid differentiation useful for quality control.
- High‑abundance compounds identified justify targeted isolation for antimicrobial and nutraceutical evaluation.
References
Fabricant, D. S., & Farnsworth, N. R. (2001). The value of plants used in traditional medicine for drug discovery. Environmental Health Perspectives, 109(suppl 1), 69–75.
Fawze, K. H. (1987). The Medical Plant. University of Alexandria.
Harborne, J. B. (1998). Phytochemical Methods (3rd ed.). Chapman & Hall.
Lee, S. B., Cha, K. H., Kim, S. N., et al. (2007). The antimicrobial activity of essential oil from Dracocephalum foetidum. Journal of Microbiology, 45, 53–57.
Mathur, P., & Choudhry, M. (2009). Consumption pattern of fenugreek seeds in Rajasthani families. Journal of Human Ecology, 25, 9–12.
Pazır, F., & Alper, Y. (2016). Carob (Ceratonia siliqua L.) fruit and health. Akademik Gıda, 14, 333–341.
Simsek, S. (2017). Chemical composition of carob pods at different harvesting stages. Food Chemistry, 221, 191–198.
Thomas, J. E., Bandara, M., Lee, E. L., et al. (2011). Biochemical monitoring in fenugreek to develop functional food and medicinal plant variants. New Biotechnology, 28, 110–117.
Anderson, R. A., Broadhurst, C. L., Polansky, M. M., et al. (2004). Isolation and characterization of polyphenol type-A polymers from cinnamon with insulin-like biological activity. *Journal of Agricultural and Food Chemistry*, 52(1), 65–70.
Basch, E., Ulbricht, C., Kuo, G., Szapary, P., & Smith, M. (2003). Therapeutic applications of fenugreek. *Alternative Medicine Review*, 8(1), 20–27.
Jayaprakasha, G. K., & Rao, L. J. M. (2011). Chemistry, biogenesis, and biological activities of Cinnamomum zeylanicum. *Critical Reviews in Food Science and Nutrition*, 51(6), 547–562.
Kim, M. J., Ryu, G. R., Chung, S. H., et al. (2005). Protective effects of D-pinitol against streptozotocin-induced diabetic renal damage in rats. *Life Sciences*, 76(3), 315–326.
Madar, Z., & Shomer, I. (1990). Polysaccharide composition of a gel fraction derived from fenugreek and its effect on starch digestion and bile acid absorption *in vitro*. *Journal of Agricultural and Food Chemistry*, 38(7), 1535–1539.
Naidu, M. M., Shyamala, B. N., Pura Naik, J., et al. (2011). Chemical composition and antioxidant activity of the husk and endosperm of fenugreek seeds. *LWT – Food Science and Technology*, 44(2), 451–456.
Ranasinghe, P., Pigera, S., Premakumara, G. A. S., et al. (2013). Medicinal properties of ‘true’ cinnamon (*Cinnamomum zeylanicum*): A systematic review. *BMC Complementary and Alternative Medicine*, 13(1), 275.
Rao, P. V., & Gan, S. H. (2014). Cinnamon: A multifaceted medicinal plant. *Evidence-Based Complementary and Alternative Medicine*, 2014, 642942.
Sharma, R. D., Raghuram, T. C., & Rao, N. S. (1990). Effect of fenugreek seeds on blood glucose and serum lipids in type I diabetes. *European Journal of Clinical Nutrition*, 44(4), 301–306.
Tetik, F., Civelek, S., & Turhan, I. (2011). Phytochemical composition and antioxidant activity of carob pod extracts. *Journal of Food Quality*, 34(3), 187–194.

